
BREAKING NEWS ITEMS [2007]

December 30, 2007; Miami, FL - A 55-year-old Sumatran orangutan, believed to be the world's oldest, has died, a Miami zoo spokesman said. "Nonja, who was born on the Indonesian island of Sumatra and lived in Miami since 1983, was found dead Saturday morning," said Ron Magill, Spokesman for the Miami Metro Zoo. "Everybody's very sad, especially with an animal like an orangutan," Magill said. "You see a lot of yourself in these animals. The great apes are our closest relatives."
An autopsy was performed that showed that Nonja suffered a hemorrhagic stroke in her brain that made her lose consciousness, vomit, aspirate into her lungs, and asphyxiate. Further testing will determine what caused the stroke in the first place. "A typical lifespan for Sumatran orangutans is [40 - 50] years," Magill said. "Nonja had slowed down in recent years because of her age. She was really a grand old dame," Magill said.
Ref.: "Florida: World's Oldest Orangutan Dies," The Los Angeles Times, p. A15 (December 31, 2007).

Drs. Stephen Coles (L) and Milan Fiala (R) with Daughter Millicent and Mother Sally after Sally
provided us with a sample of her White Blood Cells for testing.
December 19, 2007; Mrs. Sally Gappell, 108 yo, is the oldest member of the control group in the Alzheimer's Disease study at UCLA. Results comparing this control with a 73 yo Alzheimer's Disease (AD) patient demonstrate that both uptake of amyloid-beta and expression of adhesion molecules are high on control monocytes and very low in the patient monocytes. This is in agreement with our hypothesis that innate immunity against A-Beta is preserved in subjects with normal cognition but is depressed in AD patients. - - Milan Fiala, M.D., UCLA Department of Medicine.

A picture taken through a special filter in a dark room shows the cat on the Left with a red fluorescent protein that makes the animal glow in the dark when exposed to ultraviolet rays. On the right is a normal cloned cat. (Choi Byung-kil/Yonhap/ Associated Press)
December 14, 2007; Seoul, SOUTH KOREA (AP) -- South Korean scientists have cloned cats that glow red when exposed to ultraviolet rays, an achievement that could "help develop cures for human genetic diseases," The Science and Technology Ministry said. Three Turkish Angora cats were born in January and February through cloning with a gene that produces a red fluorescent protein that makes them glow in dark. One died at birth, but the two others survived, the Ministry said.
The Ministry claimed it was the first time cats with modified genes have been cloned. Scientists from Gyeongsang National University and Sunchon National University took skin cells from a cat and inserted the fluorescent gene into them before transplanting the genetically modified cells into eggs. The development means "other genes can also be inserted in the course of cloning, paving the way for producing lab cats with genetic diseases, including those of humans, to help develop new treatments," the Ministry said.
"Cats have similar genes to those of humans," said Veterinary Professor Kong Il-keun of Gyeongsang National University. "We can make genetically-modified cats that can be used to develop new cures for genetic diseases." Keitaro Kato, a geneticist at Kinki University in western Japan who has cloned fish, said the research could be significant if it eventually helps treat people with hereditary diseases.
"People with genetic disorders usually have to receive treatment throughout their lives that is very hard on them," Kato said. "If these results can help to make their lives easier, then I think it's a wonderful thing." South Korea's scientific reputation suffered a heavy blow after much-hailed stem-cell breakthroughs by scientist Hwang Woo Suk were found to be faked in late 2005. He remains on trial on fraud and other charges.
Ref.:
"Cloned Cates Glow under UV Rays," The Los Angeles Times, p. A20 (December 15,
2007).


December 8, 2007; Coles and Schulman visit Greg Fahy and Steven Harris at 21st Century Medicine and Critical Care Research, respectively.
December 7, 2007;
Published On-line December 6, 2007;
Submitted on October 23, 2007; Accepted on November 26, 2007
"Treatment of Sickle Cell Anemia Mouse Model with iPS Cells Generated from Autologous
Skin"
by
Jacob Hanna [1], Marius Wernig [1], Styliani Markoulaki [1], Chiao-Wang Sun [2],
Alexander Meissner [1], John P. Cassady [3], Caroline Beard [1], Tobias Brambrink [1],
Li-Chen Wu [2], Tim M. Townes [4*], and Rudolf Jaenisch [3*]
1. The Whitehead Institute for Biomedical Research
Cambridge, MA 02142; USA
2. Department of Biochemistry and Molecular Genetic
University of Alabama at Birmingham
Schools of Medicine and Dentistry
Birmingham, AL 35294; USA
3. The Whitehead Institute for Biomedical Research
Cambridge, MA 02142, USA
MIT Department of Biology
Cambridge, MA 02142; USA
4. MIT Department of Biology
Cambridge, MA 02142; USA
* To whom correspondence should be addressed.
Tim M. Townes, E-mail:
ttownes@uab.edu
Rudolf Jaenisch, E-mail:
jaenisch@wi.mit.edu
It has recently been demonstrated that mouse and human fibroblasts can be reprogrammed into an embryonic stem cell like state by introducing combinations of four transcription factors. However, the therapeutic potential of such induced pluripotent stem (iPS) cells remained undefined. By using a humanized Sickle Cell Anemia mouse model, we show that mice can be rescued after transplantation with hematopoietic progenitors obtained in vitro from autologous iPS cells. This was achieved after correction of the human sickle hemoglobin allele by gene-specific targeting. Our results provide proof of principle for using transcription factor induced reprogramming combined with gene and cell therapy for disease treatment in mice. The problems associated with using retroviruses and oncogenes for reprogramming need to be resolved before iPS cells can be considered for human therapy.
Refs.:
1. Gautam Naik, "Stem-Cell Research Boosts Hopes for Sickle-Cell Patients," The Wall
Street Journal, p. B6 (December 7, 2007).
The mice had previously suffered from rapid breathing, low body weight, and low blood counts.
After treatment, their condition improved. "It was striking," said Rudolf Jaenisch of the
Whitehead Institute for Biomedical Research and MIT in Cambridge, MA.
2. Karen Kaplan, "Stem-Cell Method Finds Cure," The Los Angeles Times, pp. A1, 22
(December 7, 2007).
"Due to rejection problems, only about five percent of human sickle-cell patients are able to
identify a histocompatible donor for a bone-marrow transplant that could cure the affliction."
3. AP, "Scientists Cure Mice With Sickle Cell," The New York Times
(December 7, 2007).
4. Brian Vastag, "Sickle Save: Skin Cells Fix Anemia in Mice," Science News, Vol.
172, No. 23, p. 355 (December 8, 2007).

One of the breakthrough teams works in the lab of Prof. James Thomson, here with Lab Manager
Jessica Antosiewicz.
November 20, 2007 (CNN) -- In a pair of landmark studies, two groups of scientists announced Tuesday that they have reprogrammed human skin cells to act like embryonic stem cells, whose potential to mature into any other kind of cell in the body may ultimately prove key to curing a number of diseases.
Researchers have long considered research into embryonic stem cells as a promising avenue to replacing certain disease-damaged cells. For example, diabetes results when the pancreas cells that produce insulin are damaged or die. If those cells could be replaced by healthy cells, the disease might be cured. The work is also promising in the areas of Parkinson's Disease and spinal-cord injury.
But the destruction of embryos inherent in such work has raised a raft of ethical, legal, and political questions. Tuesday's announcement may allow scientists to sidestep those concerns.
"This is the beginning of the end of the controversy," researcher James Thomson, of the University of Wisconsin-Madison, told reporters in a conference call. Thomson, who wrote one of Tuesday's papers, developed the first human embryonic stem-cell line almost a decade ago.
Both papers reported a new, simple method that reprograms human skin cells to act like embryonic stem cells.
In the other paper, published in the on-line journal Cell, a group led by Shinya Yamanaka of Kyoto University reported that inserting four genes into human skin cells reprogrammed them into what scientists call "induced pluripotent cells" (iPS) -- which look and act like stem cells.
The cells were taken from a 36-year-old woman's face and a 69-year-old man's connective tissue.
Studying the differences in the techniques used by the two groups may help researchers figure out how to turn back the developmental clock, Yamanaka said. But he said it is unclear how closely the new cells mimic stem cells. "Further studies are required to determine whether iPS cells can replace human embryonic stem cells," he said.
Last year -- using mouse cells -- Yamanaka proved that this process of reprogramming "adult" stem cells to an embryonic state was possible. The paper from the study led by Thomson, published in the on-line journal Science, says the group reported that it reprogrammed human skin cells from a baby's foreskin. Two of the genes Thomson used to convert the cells are different from those used by Yamanaka. Despite the scientists' excitement, much work remains to be done before the advances can be used to treat disease in people, they said.
In both studies, the induced cells that were formed contained several copies of viruses that were used to insert the genes into the skin cells. "Those could easily lead to mutations that might cause tumors in tissues grown from the cells," an Editorial accompanying the Science report said. But researchers said further advances may overcome that drawback.
"This is an exciting advance" said Dr. John Gearhart, a stem-cell researcher at the Johns Hopkins School of Medicine, who was not involved in either study. "This opens up a huge field for researchers." However, Gearhart said, scientists "shouldn't abandon embryonic stem cell research" because the safety of the new cells has to be established.
In a written statement, the White House said President Bush was "very pleased" with the reports. "By avoiding techniques that destroy life, while vigorously supporting alternative approaches, President Bush is encouraging scientific advancement within ethical boundaries." Bush has twice vetoed Bills that would have eased restrictions on the use of Federal funds for research involving embryonic stem cells.
Stem cell research is not banned. Private research using embryonic stem cells is allowed, but Federal money -- the largest single source of funding for such work -- is limited to research involving embryonic stem cells that were created before August 2001, when Bush announced that Federal funds could be used only for work on lines already in existence. Some researchers say the cells left available are not useful for current work.
November 20, 2007; Published on-line today in the Reports Section of AAAS
Science...
Submitted on October 9, 2007; Accepted on November 14, 2007
"Induced Pluripotent Stem Cell Lines Derived from Human Somatic Cells,"
Junying Yu [1*], Maxim A. Vodyanik [2], Kim Smuga-Otto [1], Jessica Antosiewicz-Bourget
[1], Jennifer L Frane [3], Shulan Tian [4], Jeff Nie [4], Gudrun A. Jonsdottir [4], Victor Ruotti ,
Ron Stewart , Igor I. Slukvin [5], James A. Thomson [6*]
1. Genome Center of Wisconsin, Madison, WI 53706-1580; USA.
Wisconsin National Primate Research Center
University of Wisconsin-Madison, Madison, WI 53715-1299; USA.
2. Wisconsin National Primate Research Center
University of Wisconsin-Madison, Madison, WI 53715-1299; USA.
3. Genome Center of Wisconsin, Madison, WI 53706-1580; USA.
4. WiCell Research Institute, Madison, WI 53707-7365; USA.
5. Wisconsin National Primate Research Center
University of Wisconsin-Madison, Madison, WI 53715-1299; USA.
Department of Pathology and Laboratory Medicine
University of Wisconsin-Madison, Madison, WI 53706; USA.
6. Genome Center of Wisconsin, Madison, WI 53706-1580; USA.
Wisconsin National Primate Research Center
University of Wisconsin-Madison, Madison, WI 53715-1299; USA.
Department of Anatomy, University of Wisconsin-Madison
Madison, WI 53706-1509; USA.
To whom correspondence should be addressed:
Junying Yu , E-mail:
jyu@primate.wisc.edu
James A. Thomson , E-mail:
thomson@primate.wisc.edu.
Refs.:
(Note: The four main US print newspapers [1, 3, 5-6] all had this same story printed on
Page 1, so clearly everybody thought it was among the most important stories of the day.)
1. Karen Kaplan, "Stem Cell Milestone Achieved: Scientists Coax Mature Human Cells To
Behave Like Embryonic Ones, a Step That May Bridge the Ethical Divide," The Los Angeles
Times, pp. A1,15 (November 21, 2007).
2. Ricardo Alonso-Zaldivar, "Science Can't Escape Debate," The Los Angeles Times, p.
A15 (November 21, 2007).
3. Gina Kolata, "Scientists Bypass Need for Embryo To Get Stem Cells: Method Using Human
Skin Is Seen as Defusing the Debate over Ethics," The New York Times, pp. A1,2,21
(November 21, 2007).
4. Sheryl Gay Stolberg "Advance on Stem Cells Equalizes Debate," The New York
Times, p. A21 (November 21, 2007).
5. Gautam Naik, "Advance in Stem-Cell Work Avoids Destroying Embryos," The Wall
Street Journal, pp. A1,15 (November 21, 2007).
6. Elizabeth Weise, "Stem Cell Discovery Hailed As Milestone: Technique Doesn't Destroy
Embryos; Obstacles Remain," USA Today, p. 1A (November 21, 2007).
7. World Science, "Breakthrough May Let Scientists Make Stem Cells on
Demand" (November 20, 2007).
8. NPR Radio, The World (November 20,
2007).
9. Richard Hayes, Executive Director of The Center
for Genetics and Society in Oakland, CA, OpEd Piece, "Beyond the Embryo Fight,"
The Los Angeles Times p. A39 (November 22, 2007).
Our former enemies wish to find common ground on consequential issues, now that they believe
that the ease of reprogramming of human skin cells makes the issue of harvesting human
Embryonic Stem Cells moot. However, see the next reference [10].
10. Insoo Hyun, Konrad Hochedlinger, Rudolf Jaenish, and Shinya Yamanaka, "New Advances
in iPS Cell Research Do Not Obviate the Need for Human Embryonic Stem Cells," Cell
Stem Cell, Vol. 1, p. 367-8 (October 2007).
So, I expect that the contest with our enemies will continue for some time in the future.
11. Alice Park, "Life after Embryonic Stem Cells," Time Magazine, Vol. 170, No. 23,
pp. 63-5 (December 3, 2007).
"This stem-cell breakthrough is the [Time Magazine] scientific bulletin of the year
[2007][out of 40 other notable science stories that were also covered in this issue]."
12. Andrew Pollack, "After Stem-Cell Breakthrough, the Real Work Begins," The New York
Times, pp. D1,4 (November 27, 2007).
13. Editorial, "Research on Human Embryonic Stem Cells Must Go On," Nature, Vol.
450, pp. 585-6 (November 29, 2007).
14. Patrick Barry, "Hold the Embryos: Genes Turn Skin into Stem Cells," Science News,
Vol. 172, No. 21, p. 323 (November 24, 2007).
15. Martin Fackler, "Risk Taking Is in His Genes," The New York Times, pp. D1,4
(December 11, 2007).
Interview with Shinya Yamanaka, M.D., Ph.D. of Kyoto University who maintains a two-person
lab at the University of California in San Francisco for the purpose of hESC comparisons, which
he is not permitted to do in Kyoto due to government restrictions.
16. Steve Frnst, "Editorial: Stem Cells Bust Out," Bioscience Technology, Vol. 31, No.
12, p. 6 (December 1007).




November 16, 2007; 42 persons attended the Longevity Workshop at the Embassy Suites Hotel on East Imperial Avenue in El Segundo, CA from [2:00 - 5:30] PM with presentations by Drs. L. Stephen Coles, Aubrey de Grey, Michael R. Rose. David Kekich chaired a panel discussion at the end of the day which was hosted by Susan and Bruce Klein and Mr. Elon Musk.
A video of the three-hour workshop presentations is planned to be posted on the Internet in the near future.

November 15, 2007; Prof. David Anderson introducing one of the speakers at the CIRM- sponsored Stem Cell Symposium on Thursday at the Beckman Institute at CalTech in Pasadena, CA. Invited Speakers included Drs. Ihor Lemischka of Mt. Sinai Medical Center in New York, Utpal Banerjee of UCLA, Benjamin Deverman of CalTech, Gay Crooks of Children's Hospital Los Angeles, Tatjana Sauka-Spengler of CalTech, Michael Kahn of Keck/USC, Agnes Lukaszewicz of CalTech, and Owen Witte, M.D. Director of the Broad Institute for Stem Cell Biology and Medicine at UCLA. The meeting was hosted by Prof. Paul Patterson of the CalTech Biology Department.
Dr. Lemischka, Ph.D., Director of the Black Family Stem Cell Institute and Lillian and Henry M. Stratto Professor in the Department of Gene and Cell Medicine at Mount Sinai Medical Center in New York City provided an excellent overview of stem-cell biology from a "Systems Biology" point of view. Following the pioneering work of Prof. Eric H. Davidson with Sea Urchins at CalTech, networks of pleuripotent stem-cell genes in mice (with multiple connections for up-and-down regulation) that activate murine embryogenesis program(s) are now in the process of being uncovered. Such genes are now known to include not only the well- known Oct4, Sox2, cMyc, and Nanog gene-families but the Klf4, Erk1/2, Esrrb, Tbx3, and Dppa4 gene-families as well. RNA techniques [using RNAi's (inhibitory) and shRNA's (short hairpin for knockout of gene expression)] are being used to reveal the types of connections within each network. Many of these same genes are conserved for both mouse and human species. 242 up-regulating genes and 30 down-regulating genes have recently been identified. Actin-dependent Chromatin Remodeling Complexes are also important (See J. Wang, et al., Nature (2006)).
According to Dr. Lemischka (and one of his colleagues at Princeton University, Ronald Weiss), "We are now ready to start designing artificial Embryonic Stem-Cell Circuits to test our models." This would include introducing genetic components for (1) toggle switches (to turn gene expression on or off as a function of time [not necessarily chronological time but sequentially timed events]); (2) rheostats (for up-and-down regulation); and (3) oscillators (like a Nanog Oscillator, for example). This would help speed the process of understanding embryogenesis from the formation of a zygote, to a blastula, to gastrulation (formation of three tissue types: mesoderm, endoderm, and ectoderm) and to help validate our network models. Having a standard set of instrumentation "probes" such as engineers use to trouble-shoot electrical circuits, like voltmeters, ohmeters, ammeters, etc., but which could be spliced-in at the DNA level and "read out" during gene expression without affecting the program of biological development would really make a big difference in our progress in the next ten years. Standard breakpoints could be spliced into the genome in much the same way that compiler-language software debugging tools operate, so that embryogenesis could be temporarily "frozen" and all the values of current variables could be interrogated and read out, like mRNA concentrations for example. Also, I suspect that if this sort of work were to be done at industrial-strength with thousands of biological engineers working on it at a government-run lab, instead of with a small handful of post docs associated with university-based labs, ten years of work could be accomplished in five years. This would not only speed the discovery of new interventions in cancer and other chronic age-related diseases but help in the rejuvenation of old tissues in general. This would be the beginning of a true attack on reversing the aging process itself!

November 14, 2007; New York, NY (AP and CNN) -- Gleaning stem cells from cloned monkey embryos, as a team of Oregon researchers has done, is an impressive step. But it probably won't lead to medical treatments any time soon.
Scientists merged skin cells of a Rhesus macaque male with unfertilized monkey eggs that had the DNA removed.
One hurdle is ethical and political. Human embryos have to be destroyed to produce stem cells. That has aroused opposition to human embryonic stem-cell research, and it led the Bush Administration to restrict Federal funding for it. Scientists say that has slowed science in this effort.
Another hurdle is the inefficiency of the process. Even if the method described by scientists Wednesday works in humans, it would demand too much of a precious resource -- women's unfertilized eggs. The promise of producing stem cells by cloning is that they can be genetically matched to a particular patient. So theoretically, doctors should be able to transplant tissue created from them into that person without tissue rejection. And presumably, such transplants could help treat such conditions as diabetes and spinal-cord injury.
The process used in the new experiment is "quite inefficient," Shoukhrat Mitalipov of the Oregon National Primate Research Center in Portland, OR told reporters Wednesday. He and his colleagues reported getting two batches of stem cells that required using about 150 monkey eggs apiece. That's far too many if one hopes to use human unfertilized eggs, which are cumbersome to obtain from women. If further work can get that down to maybe [5 - 10] eggs per stem cell batch, "we will be closer to clinical applications," Mitalipov said. "I am quite sure it will work in humans," he added. But then there's another issue -- showing that such stem cells really can be used to treat diseases safely. Mitalipov said he plans to do diabetes studies in monkeys. For now, he and other scientists said, the new work is valuable for showing that stem cells can be produced through cloning in monkeys. It's been done in mice, but scientists had long been frustrated in their attempts in primates, where the research would be more relevant to humans.
The new work was published on-line Wednesday by the science journal Nature. The success was reported earlier this year at a research meeting in Australia, where it received limited media coverage. The results were given new attention Tuesday by a London newspaper, The Independent.
Dr. George Daley of the Harvard Stem Cell Institute, who was familiar with the work, told The Associated Press in an E-mail that it was a "a very important demonstration" that the process is feasible in primates.
Mitalipov's team merged skin cells of a 9-year-old rhesus macaque male with unfertilized monkey eggs that had the DNA removed. The eggs, now operating with DNA from the skin cells, grew into early embryos in the laboratory. Stem cells were recovered from these embryos. The researchers have applied for a patent on their procedure.
Mitalipov said separate experiments obtained monkey stem cells from a different process called parthenogenesis, in which an egg grows into an early embryo without any genetic contribution from a male. The stem cells were genetic matches to the females that produced the eggs, he said, and early experiments suggest stem cells derived this way may someday prove useful for treating disease in women.
Nature took the unusual step of asking a separate group of scientists to verify Mitalipov's cloning results, and it published the verification along with Mitalipov's paper. In an E-mail, the journal cited the highly publicized 2004 fraud that came out of South Korea, where researchers led by Hwang Woo Suk claimed to have produced stem cells from a cloned human embryo. The journal said the research to verify Mitalipov's findings didn't signal mistrust, but noted that questions would likely be raised, and "we view this as a relatively straightforward way of putting these questions to rest."
The verification study, by David Cram and others at the Monash University in Australia, used DNA analysis of the male macaque, the two monkeys whose eggs were used to create embryos, and the stem cells. The result "demonstrates beyond any doubt" that the stem cells came from cloned embryos, the Australians wrote in their Nature paper.
______________________________________________________________Nature advance on-line publication November 14, 2007; Published on-line November 14, 2007
"Primates Join the Club"
by
Ian Wilmut and Jane Taylor
Abstract:
Researchers have achieved the testing goal of generating embryonic stem cells from the cells of an adult primate. The procedure used could provide insights into a variety of diseases, if it can be applied in humans.
Nature advance on-line publication November 14, 2007; Published on-line November 14, 2007"Producing Primate Embryonic Stem Cells by Somatic-
Cell Nuclear Transfer"
by
J. Byrne, D. Pedersen, L. Clepper, M. Nelson, W. Sanger, S. Gokhale, D. Wolf, and S.
Mitalipov
Abstract:
Derivation of embryonic stem (ES) cells genetically identical to a patient by somatic cell nuclear transfer (SCNT) holds the potential to cure or alleviate the symptoms of many degenerative diseases while circumventing concerns regarding rejection by the host immune system. However, the concept has only been achieved in the mouse, whereas inefficient reprogramming and poor embryonic development characterizes the results obtained in primates. Here, we used a modified SCNT approach to produce rhesus macaque blastocysts from adult skin fibroblasts, and successfully isolated two ES cell lines from these embryos. DNA analysis confirmed that nuclear DNA was identical to donor somatic cells and that mitochondrial DNA originated from oocytes. Both cell lines exhibited normal ES-cell morphology, expressed key stem-cell markers, were transcriptionally similar to control ES cells and differentiated into multiple cell types in vitro and in vivo. Our results represent successful nuclear reprogramming of adult somatic cells into pluripotent ES cells and demonstrate proof-of-concept for therapeutic cloning in primates.
Refs.:
1. Gina Kolata, "Scientists Use Monkey Clones for Stem Cells," The New York Times,
pp. A1,22 (November 15, 2007).
2. Karen Kaplan and Thomas A. Maugh, II, "Cloned Monkey Embryos a Giant Step' for Stem
Cells: A Breakthrough in Primates Could Help Make the Process Possible in Humans," The
Los Angeles Times, pp. A1,21 (November 15, 2007).
3. Elizabeth Weise, "A First" Stem Cell Lines Made from Embryos of Primates," USA
Today, p. 7D (November 14, 2007).
4. Gautam Naik, "Tailor-Made Stem Cells Move Step Nearer Reality: Monkey Embryos Cloned
by Scientists in Oregon; Medical Goal Is Distant," The Wall Street Journal, pp. A1, D4
(November 15, 2007).
5. CNN (November 14, 2007).
6.
CNN (January 13, 2000).

November 1, 2007 ( Science Daily) - - The DNA of a 4-year-old Abyssinian cat
named Cinnamon, whose well-documented lineage can be traced back several
generations to Sweden, has been sequenced. Cinnamon is one of several mammals that are
currently being analyzed using "light" (two-fold) genome sequence coverage. To make sense of
Cinnamon's raw sequence data, a multi-center collaboration of scientists leveraged information
from previously sequenced mammalian genomes as well as previous gene-mapping studies in the
cat. In doing so, they found that Cinnamon's sequences spanned about 65 percent of the
euchromatic (gene-containing) regions of the feline genome.
The similarity between the cat genome and six recently completed mammalian genomes (human,
chimpanzee, mouse, rat, dog, and cow) allowed the scientists to identify 20,285 putative genes in
the cat genome. The comparison also revealed hundreds of chromosomal rearrangements that
have occurred among the different lineages of mammals since they diverged from a diminutive
ancestor that roamed the earth among the dinosaurs some 100 million years ago.
The genome sequence analysis is certainly expected to lead to health benefits for domestic cats,
90 million of which are owned by Americans alone, according to The Humane
Society. But the domestic cat also serves as an excellent model for human disease, which is
one reason why the National Human Genome Research Institute (NHGRI) initially
authorized the cat genome sequencing project three years ago.
Domestic cats possess over 250 naturally occurring hereditary disorders, many of which are
similar to genetic pathologies in humans. For example, Cinnamon's pedigree carries a genetic
mutation that causes Retinitis Pigmentosa, a degenerative eye disease that can lead to blindness.
In humans, Retinitis Pigmentosa affects 1 in 3,500 Americans. The domestic cat also serves as an
excellent model for human infectious diseases, including HIV/AIDS. Feline Immunodeficiency
Virus (FIV) is a genetic relative of Human Immunodeficiency Virus (HIV), which causes
AIDS.
Using the cat genome sequence data, the researchers identified several hundred thousand
genomic variants (known as SNP's, DIP's, and STR's), which can be used to determine the
genetic basis for common hereditary diseases. The scientists havealready used these variants to
identify the causative gene for Cinnamon's Retinitis Pigmentosa (they published a paper
describing this study in the May/June, 2007 issue of the Journal of Heredity). These
variants will also be useful for parentage testing, forensic analysis, and studies of evolution,
including the reconstruction of domestication processes, fancy breed development, and
ecological adaptation among the great roaring cats.
The researchers also analyzed the feline genome for interesting features such as microRNA's,
Numts (pronounced "new mights" -- nuclear genomic fragments that migrated to cat
chromosomes from mitochondria), and a vast sea of selfish DNA-like repetitive elements. The
repetitive elements included scores of genomic stretches from historic retroviruses, some with
known links to cancer.
About Cinnamon
Cinnamon is a shy cat. While the other cats in Dr. Kristina Narfstrom's ophthalmology research
center at the University of Missouri College of Veterinary Medicine vie for the attention of
veterinary medical students and technicians, Cinnamon is more reticent, preferring to sit quietly
and watch the other cats play and carouse.
Future Research
Dr. Narfstrom predicts that the feline genome will rapidly shed new research light on a number
of cat and human medical problems including virus-mediated diseases, respiratory problems,
cancers, AIDS, immunity problems, Muscular Dystrophy, Polycystic Kidney Disorder, and Dr.
Narfstrom's research specialty, retinal eye diseases. Cats and humans share about 250 genetic
diseases.
The Cat Genome Project is based at the National Cancer Institute (Frederick, MD). Cinnamon
lives in a cat colony maintained at the University of Missouri-Columbia. The sequencing data
were generated by Agencourt Bioscience Corporation (Beverly, MA). A report that appears in the
scientific journal Genome Research details the first assembly, annotation, and comparative
analysis of the domestic cat genome ( Felis catus).


October 8, 2007; Stockholm, SWEDEN; Ground-breaking work with genes has earned three men this year's Nobel Prize for Medicine. Briton Sir Martin Evans and naturalized Americans Oliver Smithies and Mario Capecchi (of the University of Utah and a former student of James Watson) were rewarded by the Nobel Committee in SWEDEN. Using mice, they have developed a technique known as gene targeting. Capecchi said, "Gene targeting is a method for generating changes in DNA, that is genes, and then being able to evaluate what that change does to the life history of that mouse. For example, if a little finger disappeared, then we would know that that gene was important for making little fingers."
[Editor's Note: The institute that funded most of Mario Capecchi's work rejected his initial grant proposal on the grounds that "it would never work!"]
The process has been used to genetically engineer mice, which are bred with serious medical conditions such as cancer. The rodents' genes are then turned on and off to determine their effect on diseases and physiological development. Receiving their award in December, the winners will share a check for over one million Euros [US$1.54 million].
Refs.:
1. "The Nobel Prize in Medicine Was Awarded to Two US Scientists and a Briton for Their
Work on Gene Targeting, a Technique That Lets Researchers Manipulate Mouse Genes To Study
Their Effect on Disease," The Wall Street Journal, p. A1 (October 9, 2007).
2. Thomas H. Maugh, II, "Three Win Nobel for Gene Work," The Los Angeles Times,
pp. A1,10 (October 9, 2007).
3. Lawrence K. Altman and Nicholas Wade, "Three Share Nobel in Medicine for a Breakthrough
Gene Technique," The New York Times, pp. A1, D3 (October 9, 2007).
4. Steve Sternberg, "Scientists Win Nobel for 'Designer Mice', USA Today, p. 9D
(October 9, 2007).
5. AP, "Mice Gene Trio Win Medicine Nobel,"
CNN (October 8, 2007).
October 6-7, 2007

Prof. Ralph Merkle, Ph.D., Georgia Institute of Technology, "Molecular
Nanotechnology"

Dr. Michael West, Ph.D., President and Chief Science Officer of Advanced Cell
Technology (ACT), San Francisco, CA, "Immortal Cells: The Prospect of Regenerative
Medicine"

Dr. Aubrey de Grey, Ph.D., Cambridge University, UK; "Is it Politically Safe for a
Biologist to Endorse Anti-Aging Medicine?"

Dr. Steven B. Harris, M.D., President and Director of Medical Research at Critical
Care Research and 21st Century Medicine in Rancho Cucamonga, CA, "Rapid Hypothermia
Induction Methods and Brain Oxygen Requirements in Resuscitation"

Dr. Chris Heward, Ph.D., President of Kronos Science Laboratory, Phoenix, AZ, "The Kronos
Longitudinal Aging Study: The Measurement of Human Aging"
All of these speakers were also LA-GRG speakers at one time. Still other GRG speakers in attendance were Drs. Joe Schulman, David Larocca, and Steve Coles.
October 3-4, 2007;

Prof. G. Steven Burrill, U.C. San Francisco, CA




September 25, 2007; Prof. James D. Watson's Genome full Sequence has now been published on the Cold Spring Harbor Laboratory website. However, his APO E4 alleles have been blocked out intentionally, as he chose not to know his predilection for Alzheimer's Disease.
Dr. Watson's latest book Avoid Boring People: Lessons from a Life in Science has
now been published.

Although Jim's now nearly 80, he's still as sharp and active as he ever was.

September 15, 2007; Australian Biologist Alan O. Trounson of Monash University in Melbourne AUSTRALIA has been tapped to replace Dr. Zach W. Hall as President of the California Institute of Regenerative Medicine (CIRM). His annual salary will be $475,000.
In a related news item, California Treasurer Bill Lockyer has set September 27th as the date for the first offering of guaranteed taxable general-obligation bonds to finance stem-cell research under Prop. 71 in the amount of $250 million. The proceeds from the initial bond sale will be used to repay loans extended by Gov. Schwarzenegger over the past three years while CIRM was forced to undergo three rounds of litigation in California courts.
Refs.:
1. Mary Engel, "Stem-Cell Pioneer to Lead Institute," The Los Angeles Times, p. B3
(September 15, 2007).
2. Constance Holden,"Regenerative Medicine: Australian Scientist To Head California Stem Cell
Effort," Science, Vol. 317, No. 5845, p. 1667 (September 21, 2007).
3. "Q&A: King of the Stem Cells," Nature, Vol. 449, No. 7161, p. 385 (September 27,
2007).
On September 14th, The California Institute for Regenerative Medicine (CIRM)
appointed Australian Biologist Alan Trounson as its new President. The CIRM has lacked a
permanent Director since Neuroscientist Zach Hall departed in April, amidst rumors of tension
between himself and Robert Klein, Chair of the $3-billion Agency's Board.
September 12, 2007; (AP) - - The number of US deaths rose in 2005 after a drop in 2004, while heart disease and stroke killed fewer people although cancer deaths increased. Life expectancy inched up to 77.9 years [1]. In a related story, fewer children are dying due to improved sanitation and decreasing poverty according to the UN [2].
For a historical perspective, consider the following table...
Average Life Expectancy (Years)
Cave Man 18
Ancient Egypt 25
1400 Europe 30
1800 Europe 37
1900 USA 49
2002 USA 78
Refs.:
1. AP, "Life Expectancy in U.S. Hits a High," The Wall Street Journal, pp. A1,
D6 (September 13, 2007).
2. Jia-Rui Chong, "Fewer Children Dying, U.N. Says: Global Health Chief Credits Improved
Sanitation, Decreasing Poverty, and Health Programs That Provide Vaccinations," The Los
Angeles Times, p. A12 (September 13, 2007).
3. "Life Expectancy in U.S. Up a Bit, The Los Angeles Times, p. F4 (September 17,
2007).

September 3, 2007; Rockville, MD, Dr. J. Craig Venter has published his personal genome in diploid format (at the 96 percent level) in the journal PLoS Biology[1, 2]. Genetic diversity among humans has proven to be greater than previously thought. Dr. Venter will publish his autobiography next month [3]. Dr. Venter held out the prospect of a $1,000 genome for the public in just a few years [7].
Refs.:
1. Samuel Levy, et al., "The Diploid Genome Sequence of an Individual Human,"
PloS Biology, Vol. 5, No. 10, p. e254 (September 4, 2007).
2. J. Craig Venter Institute (JCVI),
Press Release
(September 3, 2007).
3. J. Craig Venter, A Life Decoded (400 pages; Viking Press, New York; October 18,
2007).
4. Nicholas Wade, "In the Genome Race, the Sequel Is Personal: Using His Own DNA, a
Pioneering Biologist and His Team Have Decoded a New Version of the Human Genome,"
The New York Times, pp. D1, 4 (September 4, 2007).
5. "Letters: A Biologist and His Genome," The New York Times, p. D4 (September 11,
2007).
6. Clive Cookson, "DNA Pioneer Reveals His Own Genetic Secrets," Financial Times,
p. 2 (September 4, 2007).
7. Interview, CNN International TV (9:15 AM PDT; September 4, 2007; TRT = 4:36
min.).
8. Brian Vastag, "The Venter Decryption: Biologist Decodes His Own Genome," Science
News, Vol. 172, No. 10, pp. 147-8 (September 8, 2007).
9. Heidi Ledford, "All about Craig: The First 'Full' Genome Sequence, Revealing That the
Genetic Pioneer Has Wet Earwax, and more..." Nature, Vol. 449, No. 7158, p. 6
(September 6, 2007).
10. Wired Geekipedia, p. 60 (Supplement to Wired Magazine, Vol. 17, No. 10
(October 2007).
The Archon X Prize sponsored by Stewart Blusson, J. Craig Venter, and others is to
sequence 100 human genomes in a ten-day period by October 2013 at US$1,000.00 each or less.
The value of the prize for the first to do this will be $10 million.
11. "The First Diploid Sequence of an Individual Human: The Highly Accurate Sequence
Suggests That Our Genetic Code Is Five Times As Variabcle As We Thought," Technology
Review, Vol. 110, No. 6, p. 92 (November/December 2007).

Yone Minagawa, 114, is congratulated by the staff at the Keiju Nursing Home at
Fukuchi (town) in Fukuoka Prefecture (state), Southwestern JAPAN on January 30, 2007.
Click on her photo for an Obituary from The Los Angeles Times.
August 14, 2007; Fukuoka, JAPAN (Agence France-Presse) -- The world's oldest person, a Japanese woman who counted eating well and getting rest as her hobbies, has died aged 114, a news report says. Yone Minagawa, a widow who lived in a nursing home but was still sprightly late in life, died "of old age" yesterday, Kyodo News reported.
There was no immediate answer to a telephone call placed to city hall in her town in southern Fukuoka prefecture. Born on January 4, 1893, Mrs. Minagawa was already in her 50's when Japan surrendered in World War II. She had been certified as the world's oldest person by the Guinness Book of World Records after Emma Faust Tillman, the daughter of freed American slaves, died in January. Despite her advanced age, Mrs. Minagawa was said to enjoy eating sweets and counted eating well and getting a good night's sleep as the secrets of her longevity.
Her nursing home said Mrs Minagawa had celebrated becoming the world's oldest person earlier this year with a Western-style lunch of bread, stew, salad and a dessert. Japanese women are the world's oldest living people, in what experts attribute to a traditionally healthy diet and high standard of medical care. Their life expectancy was a record 85.81 years in 2006, according to the Government. Japanese men are the world's second oldest with a life expectancy of 78.8 second only to men in Iceland who on average live to be 79.4.
Ref.:
Mary Rourke, "Obituary: Yone Minawawa, 114; Certified As World's Oldest Living Person,"
The Los Angles Times, p. B9 (August 14, 2007).

William Alfred Seegers [October 24, 1900 - July 10, 2007]
"Richmond Man Was Drafted by Germany"
by
Chris Treadway, Contra Costa Times; 510-262-2784 or
ctreadway@cctimes.com.
July 13, 2007; Richmond, CA -- Resident Pvt. George Johnson was California's last World War I veteran when he died in August at age 112, and it seemed that was the final chapter in the story of California residents who served during The Great War. At the time, all the remaining soldiers worldwide were older than 100 years old and seemed unlikely to relocate. But in October, William Seegers left Pennsylvania to live with his daughter in -- of all places -- Richmond, CA. He not only was a veteran of the war, but he also served for "the other side."
Seegers, a reluctant draftee into the army of Kaiser Wilhelm II in 1917, died early Tuesday morning at age 106, just weeks after his veteran status had been verified. "It's kind of ironic that lightning would strike twice in the same place like that," said Mr. Robert Young, a Senior Investigator for the Los Angeles-based Gerontology Research Group. Young, who verifies claims of "Supercentenarians," interviewed Seegers in May and verified his claim of being a World War I veteran.
"It's not surprising to find another one, it's just surprising to find another one in the same city," Young said. "There's a chance there's somebody else that someone knows about." When Seegers was added to the list of known WWI veterans, there was only one other from GERMANY, although there also were three American veterans and a Canadian veteran living in the United States. All told throughout the world, Seegers made 25.
Only 17 when he was drafted, Seegers was among the youngest of the surviving veterans. He never saw active combat and survived not only the war but also childhood malnutrition, tuberculosis, and the Spanish flu.
His early experiences forged a personality that stressed independence, self-reliance, and physical activity. Seegers had a lifelong passion for the outdoors -- particularly gardening and hiking and he celebrated his 106th birthday and his arrival in California with a hike in Muir Woods National Monument in Marin County, said Daughter Virginia Harrison of Richmond. CA.
Long before the 1960's he was involved with the conservation movement and other progressive causes, and he had lived on a commune. Seegers and his second wife, the former Eleanor Teasdale, also shared a love of performing and teaching international folk dancing, an activity he started at age 14 and gave up at 94.
As his wife's health declined, Seegers was her primary caretaker until her death in 2005. He continued to live on his own in the house he built in suburban Philadelphia until coming to live with his Daughter in October. "He had a real pride in self-sufficiency," Harrison said. "He really, strongly didn't want to have to depend on someone else."
Born October 24, 1900, outside Brinkum, GERMANY, Seegers was the second of five siblings. His family was poor, and food often was scarce while he was growing up. About the time he was four, the household moved to a town in the Harz Mountains that was less hostile to the family's socialist leanings.
Tight times grew worse as GERMANY became mired in war in 1914. "The family lived through the blockade," Harrison said. "The citizens suffered through the war and starved. My Father told stories about getting one egg a week to eat."
With discretionary income nonexistent, young Seegers found a free source of recreation with an organization called Naturfreunde (Nature's Friends), a back-to-nature movement that would have a profound influence on his life, fostering a love of the outdoors. The organization, founded in 1895 and still in existence, promotes the appreciation and conservation of the natural world. "The idea was for the people, the workers, to get away for the weekend to hike and eat and recreate," Harrison said. Seegers' family was strongly opposed to the war, to the point that they left the Lutheran Church because of its support of the Kaiser's military aims.
Seegers, who left school after the eighth grade, contracted Tuberculosis at age 15. Two years later, conscripted into the German army, he was hospitalized with the Spanish flu, a victim of the pandemic that killed more people than the war itself. In an interview in May this year, "He talked about how some of his German compatriots had died of the Spanish flu, even though they were big guys and he was a little guy," recounted Young, the gerontology researcher. The worldwide pandemic killed millions, but "Getting the flu may have saved his life because it kept him out of combat," Harrison said. In fact, "He remembered that time with some fondness," she said. All who were ill were sequestered together, regardless of nationality or allegiance. "All these guys -- Italians, French, British, German made friends with each other. He realized these people were not his enemies," Harrison said.
By the time he returned to active duty, "the German government was falling apart and the Bolshevik Revolution was going on," Harrison said. "So he just walked home. He never used the word deserted, but I realize now that's what he did. He wanted to get away. The middle class were all killing each other."
Realizing the penalty for desertion -- execution -- Seegers returned to a different infantry regiment, where he served until he was discharged in April 1919. With bleak conditions and prospects in post-war Germany, Seegers set out for the United States in 1923 with an ambition of traveling the world. He arrived in New Jersey wearing lederhosen and carrying a backpack and guitar over his shoulders, Harrison said, adding that he was dismayed to find lederhosen was not a standard form of dress here.
He left the New Jersey/New York area after a year and went to live at New Llona, a progressive and socialist commune in Louisiana. It was there that he learned the printing trade that would provide his livelihood. And it was there that he met his first wife, Vinita Seward, who was working in the print shop. They married and migrated to Philadelphia, where they both found work at union jobs as newspaper typographers. In 1933 Seegers became a naturalized citizen. Born with the given name "Wilhelm," he went by William and was known to most as "Bill."
But even as he accepted his new homeland and climbed into the middle class, the couple remained outside the American mainstream. Seegers loved the personal freedom he found in the United States, but "true to his self-sufficient nature, he criticized American society as wasteful," Harrison said. At one point in the 1930s, he delivered a radio talk to the German community on the dangers posed by Adolf Hitler and the Nazis, Harrison said. The broadcast was mysteriously cut off in midaddress.
The couple had two daughters, Ramona in 1935 and Virginia in 1943, before divorcing in 1951. Seegers married Teasdale in 1952, and they bought an acre of land for $1,000 outside Philadelphia, where he built a home for which he drew the plans himself. "He made and built it from scratch," Harrison said. "He loved doing things by himself, and he was quite good at making things out of almost nothing." It was there that he indulged his love of gardening, getting up at dawn to prune trees before leaving for work.
"His two biggest passions were dancing and being outside," said Granddaughter Julian Carter. He also enjoyed drawing, calligraphy, and photography. "He loved to make concrete changes in the world, things you could see," Carter said.

Sunday, July 1, 2007; 7:00 PM PDT -- Prof. Michael B. Pesce, M.D., J.D., of the Bioethics Institute of Loyola Marymount University in Los Angeles, is both a physician and an attorney (San Diego Law School). By medical specialty, he is an anesthesiologist. This makes him especially qualified to argue the issue in Federal and state courts regarding the constitutionality of lethal injection for the death penalty, on the grounds that it is "cruel and unusual punishment" (8th Amendment), if not supervised by a qualified physician who can ascertain whether the prisoner is truly unconscious and not merely paralyzed. The reverse has been documented to have happened with the unprofessional IV administration of a lethal cocktail of three ingredients (Procedure No. 770) that are routinely employed for this procedure today ( Sodium Thiopental [fast-acting barbiturate to render the inmate unconscious], Pancuronium Bromide [to paralyze the body], and high-dose Potassium Chloride [for cardiac arrest]). This last drug would be excruciatingly painful if one were still conscious during an IV administration and were unable to alert anyone to this frightening possibility of being conscious while paralyzed within an immobile body. Curiously, Procedure No. 770 has no provision to ensure full anesthetization, as would normally be required in any surgical procedure (or even veterinary procedure). [1]
On the other hand, the American Board of Anesthesiology has instructed its physician members not to participate in any manner in state executions on the grounds that such behavior would violate the spirit of the Hippocratic Oath to "do no harm." So what shall the state to do to rectify this dilemma? Shouldn't it train and qualify paramedics just for this purpose? Neurologists are trained to establish consciousness or brain death (by EEG) in patients suffering from a Persistent Vegetative State (PVS) that can last for years. Sadly, this obvious advice is not always heeded. Indeed, several executions by unqualified personnel in certain Southern states have been found to have been to have been conducted in a highly unprofessional manner. Revising the procedure to use a fast-acting barbiturate by mouth to put the prisoner to sleep followed by inhaling Carbon Monoxide (CO) gas through a mask (in its colorless, tasteless, odorless form, not the sort that has had other odiferous compounds added by the gas-and-electric company, so that one could instantly detect its presence in the event of an accidental gas leak), Methane (CH4) gas or, even better, pure Nitrogen (100% N2) gas would make the procedure completely painless, providing painlessness were the goal that the state had in mind. Even Dr. Jack Kevorkian's method of euthanasia (or physician-assisted suicide, which is legal in the state of Oregon, but not in Michigan where he was imprisoned) has more to recommend it. We do provide an inmate with last rites by a priest and/or last wishes regarding a final meal, don't we? It makes one wonder if "public humiliation" or "revenge" are not more appropriate terms to characterize the state's true intentions rather than a pretense that shields from an unsophisticated majority any cognitive dissonance or psychological discomfort when they hear on a news broadcast that an inmate has been executed.
In such a case, one has to ask why lethal injection has been adopted as the new standard over the more traditional methods of firing squad (still an option in Utah with or without a blindfold and with or without one of the squad members having a surreptitiously pre-loaded blank round placed in advance, so that no member would know if they were really the agent that killed the victim), hanging by noose on a gallows (short drop [asphyxiation]) vs. long drop [2nd-and-3rd or 4th-and-5th cervical vertebrae snapped suddenly]), electrocution, or the gas chamber (Sulfuric Acid [H2SO4] poured through a tube onto Potassium Cyanide [KCN] pellets), if the goal is really to have the state execute its prisoners in a more dignified, humane manner. Otherwise, why not just go back to more historical methods, like drinking a poison (Hemlock, Arsenic, Strychnine, Deadly Night Shade [Belladonna/Atropine], Fox Glove [Digitalis]), crucifixion, impaling on a sharp vertical lubricated wooden spike through the rectum, stabbing by knife, slow boiling in a vat of water, garroting, "the wheel," in which limbs are systematically fractured as it rotates, drawing-and-quartering, beheading [decapitation] by a strong axeman wearing a hood, the Guillotine (invented by a French physician of the same name), an Aztec/Mayan High Priest who cuts out the victim's heart with a stone knife and places it on a Chakmul, letting their bleeding dead body roll down a very steep flight of steps to the bottom of a pyramid (human sacrifice), cannibalism (anthropophagy), drowning (water boarding), immolation [burning at the stake], being eaten by poisonous insects or bitten by venomous snakes in a viper pit, biblical stoning, excessive blood letting to syncope (fainting) by a trained phlebotomist using a lancet in the middle ages as an alleged therapy for sanguine plethora (curiously, President George Washington died this way at the hands of his well-intentioned physicians, who were treating him for a sore throat), Chinese water torture, an Iron Lady, being buried alive, a stone room that shrinks and crushes like a vice, a pit-and-pendulum as represented in Edgar Allen Poe's tales of horror, or even something as simple as asphyxiation with a plastic bag tied over the head? These all can get the job done and have an illustrious history of effectiveness, don't they? The History Channel had a one-hour TV special on this topic a year or so ago, and I may have left out some of the more exotic, gory, or vicious methods that were never sanctioned by any state but were employed by the clergy in the Spanish Inquisition against ostensible heretics.
There are two common arguments in favor of the death penalty in the minds of the American public, both of which are completely without merit: (1) it serves as a deterrent to the commission capital crimes; and (2) the state would save money by executing prisoners rather than having them take up cell space and food for a life-time of imprisonment. In response to the first argument, we note that Texas executes prisoners at a substantially higher rate than any other state by a wide margin, while Michigan outlawed capital punishment decades ago. According to this logic, the homicide rate in Michigan ought to be substantially higher than it is in Texas. Yet, empirically, we observe the opposite to be the case. So, where's the deterrence? Similarly, the same is true in Europe where the European Union has outlawed capital punishment for decades. Regarding the second argument, the legal costs of prosecuting death penalty cases on appeal far outweighs the comparatively trivial costs of daily maintenance of prisoners in jail. So, go figure the logic of this absurd proposition.
The case of capital punishment in China is especially egregious where 90 percent of all executions take place world wide. This country surreptitiously supports the sale to wealthy foreigners of organs from executed prisoners (without their consent), preparing them in advance with HLA tissue-typing and anti-coagulation (heparin) injections in a mobile van-trailer just prior of the execution to ensure that no damage is done to their "valuable" fresh organs before the transfer from the donor to the recipient is carried out by waiting surgeons. It is hard to see the difference between this state-sponsored activity and the gruesome experiments conducted on World War II concentration-camp prisoners for which Nazi-employed German doctors were charged at the Nuremberg Trials and for which they were subsequently found guilty and executed "for crimes against humanity."
According to Dr. Pesce, "Whenever the state executes a prisoner in a non-humane manner, it lessens the dignity of all of us."
Refs.:
1. Henry Weinstien, "New Lethal Injection Plan Assailed: Lawyers for Death Row Inmate
Michael Morales Say a Revised Protocol for Executing Convicts Is Even More 'Ill-Conceived'
Than Previous Versions," The Los Angeles Times, p. B3 (July 4, 2007).
2. "Tennessee: Lethal Injection Process Is Blocked," The Los Angeles Times, p. A17
(September 20, 2007).
A Federal Judge described Tennessee's new lethal injection procedures as "cruel and unusual
punishment," and blocked an execution scheduled for next week. "The new procedures do not
ensure that inmates are properly anesthetized before the lethal injection is administered," Judge
Aleta Arthur Trauger said, "which could result in a terrifying, excruciating death."
3. AP, "Rulings May Change Lethal Injection Process," The Los Angeles
Times, p. A19 (September 22, 2007).
4. David G. Savage and Henry Weinstein, "Supreme Court To Hear Lethal Injection Case:
Justices Could Ban Commonly-Used Chemical Concoctions That May Cause Dying Inmates To
Suffer," The Los Angeles Times, p. A20 (September 26, 2007).
5. Henry Weinstein, "Execution Put Off Over Lethal Injection: Alabama's Governor Grants an
Inmate a 45-Day Stay. In a Texas Case, the U.S. Supreme Court Steps in," The Los Angeles
Times, p. A28 (September 28, 2007).
6. AP, "Texas May Halt Executions," The Los Angeles Times, p. A21
(September 29, 2007).

June 21, 2007; Washington, D.C. (AP) -- Pushing back against the Democratic-led Congress, President Bush vetoed a Bill Wednesday that would have eased restraints on Federally-funded embryonic stem-cell research. "Our innovative spirit is making possible incredible advances in medicine that can save lives and cure diseases," the President told an invited audience in the East Room. "America is also a nation founded on the principle that all human life is sacred. And our conscience calls us to pursue the possibilities of science in a manner that respects human dignity and upholds our moral values."
Democrats, who had made the stem-cell legislation a top priority when they took control of the House and Senate in January, were quick to denounce the President's decision. "This is just one example of how the President puts ideology before science, politics before the needs of our families, just one more example of how out of touch with reality he and his party have become," Sen. Hillary Clinton (D., NY) a candidate for the Democratic presidential nomination, told the Take Back America conference of liberal activists Wednesday. Sen. Barak Obama, another Democratic presidential hopeful, said Mr. Bush was "deferring the hopes of millions of Americans who do not have the time to keep waiting for the cure that may save or extend lives."
To blunt criticism, Mr. Bush issued an Executive Order directing the Health and Human Services Department to promote research into cells that -- like human embryonic stem cells -- also hold the potential of regenerating into different types of cells that might be used to battle disease.
If the measure Mr. Bush vetoed would have become law, the White House said it would have compelled taxpayers for the first time in our history -- to support the deliberate destruction of human embryos. Spokesman Tony Snow said Mr. Bush's Executive Order encouraged scientists to work with the government to add research on new stem-cell lines -- that does not involve the creation, harming or destruction of human embryos -- to the list of projects eligible for Federal funding. "The President does not believe it's appropriate to put an end to human life for research purposes," Mr. Snow said. "That's a line he will not cross."
Senate Majority Leader Harry Reid is expected to schedule an override vote, but the date has not been set. Democrats, however, currently do not have enough votes to override Mr. Bush's veto.
Scientists were first able to conduct research with embryonic stem cells in 1998, the NIH says. There were no Federal funds for the work until Mr. Bush announced in 2001 that his Administration would make the funds available for lines of cells that already were in existence.
Currently, states and private organizations are permitted to fund embryonic stem-cell research, but Federal support is limited to cells that existed as of August 9, 2001. The latest Bill was aimed at lifting that restriction. The science aside, the issue has weighty political and ethical implications.
Public opinion polls show strong support for the research, and it could return as an issue in the 2008 elections. Opponents of the latest stem-cell measure insisted that the use of embryonic stem cells was the wrong approach on moral grounds -- and possibly not even the most promising one scientifically. These opponents, who applaud Mr. Bush's veto, cite breakthroughs involving medical research conducted with adult stem cells, umbilical cord blood and amniotic fluid, none of which involve the destruction of a human embryo. [*]
This was the third veto of Mr. Bush's presidency. His first occurred last year when he rejected legislation to allow funding of additional lines of embryonic stem cells -- a measure that passed over the objections of Republicans then in control. The second legislation he vetoed would have set timetables for U.S. troop withdrawals from Iraq.
__________________
The American Association for the Advancement of Science (AAAS) on Wednesday expressed disappointment with the decision by President George W. Bush to veto a landmark stem-cell research funding Bill. The measure would have lifted restrictions on Federal funding of medical research using embryonic stem cells, which proponents say would be derived from microscopic embryos left over and due to be discarded after in vitro fertility treatments. Current U.S. policy limits such funding to fewer than 20 stem-cell lines that had been created when the president first announced U.S. policy in 2001.
"Our concern is that this action has the potential to delay development of important new therapies by cutting off a promising avenue of research," said Alan I. Leshner, the Chief Executive Officer of AAAS and executive publisher of the journal Science. The President vetoed a similar Bill that passed the Senate and House of Representatives last year. This year's measure won strong bi-partisan support in the U.S. House and Senate, but fell just short of the two-thirds "supermajority" needed to override the veto.
While Bush signed a separate Executive Order to support other stem cell research, AAAS "strongly believes" that all avenues of stem cell research must be explored, the non-partisan association said a statement.
The following is the full text of the AAAS statement:
"The President has again vetoed The Stem-Cell Research Enhancement Act, which
would expand Federal support for embryonic stem-cell research. AAAS, the world's largest
general scientific society, stands with a broad coalition of Americans spanning all parties and
faiths that supports this bill.
"The scientific consensus is that embryonic stem-cell research is an extremely promising
approach to developing more effective treatments for devastating conditions like Diabetes,
spinal-cord injuries, and Parkinson's Disease. The bill would mandate that such research be
allowed to compete for Federal funding while following strict ethical guidelines.
"The Executive Order is not a substitute for The Stem Cell Research Enhancement Act. The new
approaches addressed by the Order are still in the early stages of development and appear to
already be eligible for NIH funding. AAAS strongly believes that it is only through Federal
support of diverse avenues of stem-cell research, including especially embryonic stem-cell
research, that we may better understand the potential value and limitations of each
approach.
"During his tenure the President has acknowledged that it is a critical time for the American
scientific enterprise, therefore it is disappointing that he has chosen to maintain restrictions on
such a promising area of research. AAAS will continue to support the interests of scientists and
patients in fostering medical progress."
Refs.:
1. "Bush Vetoed Legislation To Ease His Restrictions on Federal-Funding of Embryonic Stem
Cell Research. An Override Attempt Is Seen As Unlikely," The Wall Street Journal, p.
A1 (June 21, 2007).
The Senate may be one vote shy of the two-thirds mark, but the House is about 35 votes short, so
pushing an override further at this time seems futile.
2. "Donation to Stem-Cell Research Is Backed by 60 percent of Couples with Embryos Stored at
Fertility Clinics, a Survey in the Journal Science Says," The Wall Street Journal, p. A1
(June 21, 2007).
3. "Many OK with Donating Embryos," The Los Angeles Times, p. F11 (June 25,
2007).
Of 1,020 persons who responded by saying that they still had embryos in storage, 49 percent said
they were likely to donate some or all of them for research. When specifically asked about
stem-cell research, the portion willing to donate embryos rose to 62 percent.
4. Maura Reynolds, "Bush Vetoes Embryonic Stem-Cell Funding," The Los Angeles
Times, p. A12 (June 21, 2007).
5. Editorial, The New York Times, p. A24 (June 21, 2007).
6. "Poll on Federal Funding for Stem-Cell Research," CNN (June 19, 2007).
The poll asked ordinary Americans whether we should allow for Federal funding of stem-cell
research. The results were as follows:
53% Yes
41% No
6% Undecided
7. Constance Holden, "Stem-Cell Science Advances as Politics Stall," Science, Vol. 316,
No. 5833, p. 1825 (June 29, 2007).
8. "Stem Cell Veto - - Again," Scientific American, Vol. 297, No. 3, p. 36 (September
2007).
___________
* GRG Editorial Comments on the Bush Veto:
![3. Zygote formation [0 - 1] day Zyogote formation](/images/Zygote.jpg)
![4. Pre-Embryo [0 - 2] weeks (morula) Pre-Embryo](/images/PreEmbryo.jpg)
![5. Embryo [2 - 8] weeks Embryo [2 - 8] weeks](/images/Embryo.jpg)
![6. Fetus [8 - ~40] weeks Fetus [8 - 40] weeks](/images/Fetus.jpg)
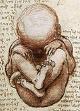
Scroll over each of the seven images with your mouse for more details. Click if you would like to see more academic details on embryology from a standard course offered by McGraw-Hill, including timing of development and human life history, along with definitions of such related terms as morula and gastrula.
It's disgusting that the White House would exploit one small aspect of the exciting mouse adult-stem-cell research reported in Cell: Stem Cell and in Nature two weeks ago [scroll down to the news item on June 6th below] to focus on the non-destruction of human embryos while ignoring the more important scientific aspect of this work (e.g., pluripotent stem cells having instant histocompatibility with the donor). This is what happens when the media allows one side to "frame the debate" in their own terms for the sake of a personal agenda while the other side gets shut out. Prof. Kathrin Plath, a stem-cell biologist at UCLA, said that Bush's order "doesn't satisfy me, because we have to study stem cells derived from fertility clinic embryos to understand what a human embryonic stem cell is all about." Plath, who was a senior author on one of the three recent papers referred to by Bush notes that her group's results haven't been reproduced with human cells yet. "Human reprogramming just might not work," she said.
Opponents of stem-cell research speak misleadingly of an "embryo" being destroyed in the harvesting of human embryonic stem cells from the inner cell mass of a blastocyst during SCNT (Somatic Cell Nuclear Transfer). One even hears disgraceful comparisons with "partial birth abortions." But no scientists we know are proposing to destroy what embryologists call an "embryo," let alone a fetus. The only tissue being destroyed would be a pre-embryo. And a pre-embryo is not an embryo. A pre-embryo is, by definition, a blastocyst (a few hundred cells)(See Image 4) starting with a fertilized egg [or oocyte] (See Images 1 and 2) or a zygote (a single cell undergoing its first mitotic division in Image 3). A blastocyst starts with a few identical totipotent cells and then begins to differentiate into a ball with well-defined "North" and "South" Poles. Trophoblastic cells will go on to form the placenta, while the inner cell mass of pluripotent cells will subsequently go on to form the embryo. A pre-embryo lasts for less than two weeks after conception. A pre-embryo floats in fluid as it slowly migrates down the Fallopian Tube when it is expelled into the uterus. It has not yet implanted in the uterine wall. It has no oxygenated blood supply of its own. Indeed, according to the latest statistics, a little over 50 percent of all pre-embryos don't even implant in the uterine lining for various reasons, mostly due to chromosomal abnormalities. They are unceremoniously expelled with the next menstrual period (a "disappointed uterus"). The woman in question is never even cognizant that she was "almost" pregnant. These lost pre-embryos are not mourned, not sought out scrupulously for baptism [or whatever "sacred rite" to avoid living permanently in "limbo" or "purgatory"], nor provided a formal grieving ritual. How could there be such a ritual? God never revealed his ways to us until recently.
By contrast, an embryo, is defined as a developing organism post-implantation in the uterine lining that begins with the formation of a placenta having its own maternal blood supply (technically, the start of a true pregnancy that will go to term in about nine months, if all goes well, resulting in the birth of an infant boy or girl). For reference, an embryo transitions into a fetus after eight weeks (See Image 5 (Embryo), and Images 6 and 7 (Fetus) above, lest there be any doubt as to what we're talking about). Thus, a pre-embryo is not human life in the sense that evangelical Christians would have us believe; certainly, a pre-embryo is human tissue with human DNA, but it is not human life or a person. A pre-embryo is only a "potential" person. (And it should be entitled to certain degree of respect as human tissue. But to demonstrate the foolishness of obfuscating or dissembling about whether all human tissues deserve equal respect -- my finger-nail clippings, my hair follicles, my blood, my saliva, etc. could be called human life, if having human DNA were the only requirement for this exalted status.) So human life (a person) does not "begin at conception," but only three or four weeks later in time. The (legal) definition of personhood is probably outside the scope of religious deliberations, although the term "ensoulment" is a related religious term that gets wide currency in theology school. However, exactly what constitutes a soul is clearly beyond the scope of the present discussion.
It's not that the President's staff doesn't understand the difference between a pre-embryo and an embryo. (I believe that they do speak English, as their native language, and they could look it up in a dictionary or on the Internet if they wanted to). It's not that they don't understand the difference between an acorn and an oak tree. (Obliterating an acorn is not the same thing as chopping down an oak tree. Ask any child.) It's not that they don't understand the abstract difference between potentiality and reality. They do. It's just that they don't want to understand. They're irrevocably locked into their religious position, and they're going to ride it into the sunset! They self-righteously accept as 'untouchably sacred' the argument that 'human life beings at conception'. For them, it is not post-14-day implantation plus a couple of weeks to allow for organogenesis to take place (Aristotle imagined that it took "40 days" [324 BCE]). For them, it's a matter of ethical certainty and absolute morality. But, we teach biomedical ethics to physicians, residents, and medical students, and we say that evangelical ethics are simply wrong. They are based on deliberate misrepresentations of modern embryology and medical facts that help their advocates frame the debate to prove their points (by intimidation). As Chris Hedges said in his book American Fascists: The Christian Right and the War on America (Free Press, New York; 2006), "The Nazis are not going to return with swastikas and brown shirts. Their ideological inheritors in America will find a mask for fascism in patriotism and the pages of the Bible." And as Al Gore said in his best-selling book The Assault on Reason (Penguin Press, New York; 2007), "The politics of fear, secrecy, cronyism, and blind faith has combined with the degradation of the public media to create an environment dangerously hostile to reason."
Thus, the only way to overcome this irreconcilable difference, as I see it, is impeachment. Is there any other way to send this man back to his ranch at Crawford, where he and his staffers can live happily ever after? We must somehow remove their cold clenched hands from the levers of power and prevent them from forcing their evangelical agenda down the throats of American citizens -- especially as the lives of so many of us are at risk from chronic age-related diseases that could be amenable to therapy using the techniques that he deliberately blocks on the grounds that "it would cross a line" (their line, not our line). We desperately need a "regime change," and it's not abroad but here at home.
-- L. Stephen Coles, Co-Founder, GRG

June 19, 2007; Until now, there has been an insurmountable barrier to cloning primate cells. Now the barrier is down, thanks to a new technique developed using rhesus monkey cells.
Refs.:
"Reprogramming Following Somatic Cell Nuclear Transfer
in Primates is Dependent upon Nuclear Remodeling"
by
S.M. Mitalipov [1,5], Q. Zhou[2], J.A. Byrne[1], W.Z. Ji[3], R.B. Norgren[4], and D.P. Wolf[1]
Human Reproduction
Oregon National Primate Research Center
Oregon Health and Science University
505 NW 185th Avenue
Beaverton, OR 97006; USA
2. Institute of Zoology
Chinese Academy of Sciences
Beijing, CHINA
3. Kunming Institute of Zoology
Kunming Primate Research Center
Chinese Academy of Sciences
Kunming, Yunnan, CHINA
4. Department of Genetics
Cell Biology and Anatomy
University of Nebraska Medical Center
NE; USA
Correspondence address:
Voice 503-614-3709
FAX: 503-533-2494
E-mail: mitalipo@ohsu.edu
Human therapeutic cloning has moved one step closer to reality. Stem cells have been extracted from cloned monkey embryos for the first time -- and if it works in monkey cells, why not in human cells too? "It's proof of principle for human therapeutic cloning," says team member Don Wolf of the Oregon National Primate Research Center in Beaverton. Wolf's colleague Shoukhrat Mitalipov announced the breakthrough on June 18th at a meeting of the International Society for Stem Cell Research in Cairns, Queensland, AUSTRALIA. The Oregon team stripped the chromosomes from 278 rhesus monkey eggs and replaced them with the nuclei of skin cells from male monkeys. They derived two stem-cell lines from 21 embryos that developed into a hollow ball of cells known as a blastocyst.
Earlier cloning attempts in monkeys used ultraviolet light as a guide while the chromosomes were being removed from eggs. The Oregon researchers believe this damaged the resulting embryos. Instead, their technique uses polarized light to visualize the egg's interior ( Human Reproduction, DOI: 10.1093/humrep/dem136).
The Oregon team has yet to show its cells have all the characteristics of embryonic stem cells. But already, other researchers are planning to try the same methods on human cells. "The primate stuff really does give us renewed hope," says Renee Reijo Pera, who heads a team working on therapeutic cloning at Stanford University in California.
4. "Monkey Clones Lead the Way," New Scientist Magazine, Vol. 194, No.
2610, p. 7 (June 30, 2007).
278 monkey eggs > 21 blastocysts > 2 stem cell lines; UV light damages chromosomes.

This photo released by the New Bedford Whaling Museum shows the tip of the bomb lance fragment patented in 1879 that was removed from the neck of a bowhead whale captured at Barrow, Alaska in May 2007. The body of the bomb lance was not recovered. The shiny scars are the result of a chain saw cut.
June 12, 2007; (AP) -- A 50-ton bowhead whale caught off the Alaskan coast last month had a weapon fragment embedded in its neck that showed it survived a similar hunt -- more than a century ago. Embedded deep under its blubber was a 3 1/2-inch arrow-shaped projectile that has given researchers insight into the whale's age, estimated between 115 and 130 years old. "No other finding has been this precise," said John Bockstoce, an Adjunct Curator of the New Bedford Whaling Museum.
Calculating a whale's age can be difficult, and is usually gauged by amino acids in the eye lenses. It's rare to find one that has lived more than a century, but experts say the oldest were over 200 years old. The whale, which was nearly half a football field long, had a bomb lance fragment lodged a bone between its neck and shoulder blade. "The fragment was likely manufactured in New Bedford, on the Southeast coast of Massachusetts, a major whaling center at that time," Bockstoce said.
It was probably shot at the whale from a heavy shoulder gun around 1890. The small metal cylinder was filled with explosives fitted with a time-delay fuse so it would explode seconds after it was shot into the whale. The bomb lance was meant to kill the whale immediately and prevent it from escaping. "The device exploded and probably injured the whale," Bockstoce said. "It probably hurt the whale, or annoyed him, but it hit him in a non-lethal place," he said. "He couldn't have been that bothered if he lived for another 100 years."
The whale harkens back to far different era. If 130 years old, it would have been born in 1877, the year Rutherford B. Hayes was sworn in as US President, when Federal Reconstruction troops withdrew from the South, and when Thomas Edison unveiled his newest invention, the phonograph.
The 49-foot male whale died when it was shot with a similar projectile last month, and the older device was found buried beneath its blubber as hunters carved it with a chain saw for harvesting. "It's unusual to find old things like that in whales, and I knew immediately that it was quite old by its shape," said Craig George, a wildlife biologist for the North Slope Borough Department of Wildlife Management, who was called down to the site soon after it was found. The revelation led George to return to a similar piece found in a whale hunted near St. Lawrence Island in 1980, which he sent to Bockstoce to compare. "We didn't make anything of it at the time, and no one had any idea about their lifespan, or speculated that a bowhead could be that old," George said.
Bockstoce said he was impressed by notches carved into the head of the arrow used in the 19th century hunt, a traditional way for the Alaskan hunters to indicate ownership of the whale. Whaling has always been a prominent source of food for Alaskans, and is monitored by The International Whaling Commission. A hunting quota for the Alaska Eskimo Whaling Commission was recently renewed, allowing 255 whales to be harvested by ten Alaskan villages over five years. After it is analyzed, the fragment will be displayed at the Inupiat Heritage Center in Barrow, Alaska.
Refs.:
1. Erin Conroy, Associated Press Writer, "19th-Century Weapon Found in Whale"
2. AP, "Whale Caught in May Was Hunted in 1890," The Los Angeles Times,
p. A20 (June 13, 2007).
[Editorial Remark: It's a shame that the bowhead whale is not a protected species so that we could discover what its maximum lifespan really is (and what their DNA sequence looks like). The whale in question might have lived a lot longer if it were not harvested for its meat. (Sigh!) Other bowheads have been found with Alaskan Eskimo hand-made harpoons in them that were even older.]

June 6, 2007; Click on the Mouse above for more details from Nature.
Ectopic expression of the four transcription factors Oct-4, Sox-2, c-Myc, and Klf-4 is sufficient to confer a pluripotent state upon the fibroblast genome, generating induced Pluripotent Stem (iPS) cells. It remains unknown if nuclear reprogramming induced by these four factors globally resets epigenetic differences between differentiated and pluripotent cells. Here, using novel selection approaches, we have generated iPS cells from fibroblasts to characterize their epigenetic state. Female iPS cells showed reactivation of a somatically-silenced X Chromosome and underwent random X-inactivation upon differentiation. Genome-wide analysis of two key histone modifications indicated that iPS cells are highly similar to ES cells. Consistent with these observations, iPS cells gave rise to viable high-degree chimeras with contribution to the germline.
These data show that transcription-factor-induced reprogramming leads to the global reversion of the somatic epigenome into an ES-like state. Our results provide a paradigm for studying the epigenetic modifications that accompany nuclear reprogramming and suggest that abnormal epigenetic reprogramming does not pose a problem for the potential therapeutic applications of iPS cells.
_________________
Editorial Comment: If this approach works in humans, it will obviate all of the ethical and religious objections to SCNT and/or the presumed exploitation indigent teenage girls who could be paid to become egg donors for research purposes. Next, we need to perform a proof-of-concept with human cells in culture starting with a simple skin biopsy. Curiously, instead of dealing with OB/GYN infertility-endocrine specialists to achieve rejuvenation therapies, we could be working with dermatologists and plastic surgeons!
Refs.:
1. Article: "Directly Reprogrammed Fibroblasts Show Global Epigenetic Remodeling and
Widespread Tissue Contribution,"
Nimet Maherali [1, 2, 6], Rupa Sridharan [3, 6], Wei Xie [3], Jochen Utikal [1], Sarah Eminli
[1], Katrin Arnold [1], Matthias Stadtfeld [1], Robin Yachechko [3], Jason Tchieu [3], Rudolf
Jaenisch [5], Kathrin Plath [3, 4], and Konrad Hochedlinger [1]. Cell Stem Cell, Vol 1,
pp. 55-70 (June 7, 2007).
[1.] Massachusetts General Hospital
Cancer Center and Center for Regenerative Medicine
Harvard Stem Cell Institute
185 Cambridge Street
Boston, MA 02114; USA
[2.] Department of Molecular and Cellular Biology
Harvard University
7 Divinity Avenue
Cambridge, MA 02138; USA
[3.] Department of Biological Chemistry
Johnson Comprehensive Cancer Center and
Institute for Stem Cell Biology and Medicine
UCLA David Geffen School of Medicine
Los Angeles, CA 90095; USA
[4.] Molecular Biology Institute
Johnson Comprehensive Cancer Center and
Institute for Stem Cell Biology and Medicine
UCLA David Geffen School of Medicine
Los Angeles, CA 90095; USA
[5.] MIT Whitehead Institute and
Department of Biology
9 Cambridge Center
Cambridge, MA 02142; USA
Corresponding author: Kathrin Plath E-mail:
kplath@mednet.ucla.edu;
Corresponding author: Konad Hochedlinger E-mail:
khochedlinger@helix.mgh.harvard.edu
2. Keisuke Okita, Tomoko Ichisaka, and Shinya Yamanaka, "Generation of Germline-Competent
Induced Pluripotent Stem Cells," Nature, Vol. 448, pp. 313-7 (July 19, 2007).
3. Marius Wernig, Alexander Meissner, Ruth Foreman, Tobias Brambrink, Manching Ku,
Konrad Hochedlinger, Bradley E. Bernstein, and Rudolf Jaenisch, "In vitro
Reprogramming of Fibroblasts into a Pluripotent ES-cell-like State," Nature, Vol. 448,
pp. 318-24 (July 19, 2007).
4. Kazutoshi Takahashi and Shinya Yamanaka, "Induction of Pluripotent Stem Cells from Mouse
Embryonic and Adult Fibroblast Cultures by Defined Factors," Cell, Vol. 126, pp. 663-
76 (August 25, 2006).
5. Gautam Naik, "Stem-Cell Advance May Skirt Ethical Debate," The Wall Street Journal
, pp. A1, B1,2 (June 7, 2007).
6. Karen Kaplan, "Skin Cell Is Made To Mimic Stem Cell: Scientists Reprogram Mouse Tail
Cells To Act Like Embryonic Lines. The Advance May Put Ethical Concerns to Rest," The
Los Angeles Times, pp. A1, 2, 18 (June 7, 2007).
7. Nicholas Wade, "Biologists Make Skin Cells Work Like Stem Cells: Avoiding Divisive
Debate: Goal Is Converting Cells into Tissues To Replace the Diseased Ones," The New
York Times, pp. A1, 2, 30 (June 7, 2007).
8. Dan Vergano, "Cells of Mice Get Radically Altered: Is It a New Way To Make Stem Cells?"
USA Today, p. 1A (June 7, 2007).
9. Rick Weiss, "Scientists Use Skin To Create Stem Cells: Discovery Could Recast Debate,"
The Washington Post, p. A1 (June 7, 2007).
10. "Loss Of Stem Cells Correlates With Premature Aging In Animal Study,"
Science Daily (June 6, 2007).
11. Jeff Cox, "Stem-Cell Industry Set to Break Out," CNN Money (June 7, 2007).
12. Constance Holden, "Stem Cells: Teams Reprogram Differentiated Cells -- without Eggs,"
Science, Vol. 316, No. 5830, pp. 1404-5 (June 8, 2007).
13. Carolyn Barry, "Biomedicine: Mouse Method Turns Skin Cells to Stem Cells," Science
News, Vol. 172, No. 2, p. 29 (July 14, 2007).
14. Shinya Yamanaka, Kyoto University, JAPAN, Nature (2007).
20 percent of the mice died of cancer brought on by two of the four genes used to reprogram skin
cells to become pluripotent stem cells.
June 6, 2007; London, UK (BBC) -- A major advance in understanding the genetics behind several of the world's most common diseases has now been reported. The landmark Wellcome Trust study analyzed DNA from the blood of 17,000 people to find genetic differences. They found new genetic variants for depression, Crohn's Disease, coronary heart disease, hypertension, Rheumatoid Arthritis, and Type-1 and -2 Diabetes. The remarkable findings, published in Nature, have been hailed as a new chapter in medical science. It is hoped they will pave the way for research into new treatments and genetic tests.
"By identifying the genes underlying these conditions, our study should enable scientists to understand better how disease occurs, which people are most at risk and, in time, to produce more effective, more personalized treatments." -- Prof. Peter Donnelly
Disease-by-Disease Findings: Q&A: Gene Discovery
The £9 million Wellcome Trust Case Control Consortium (WTCCC) involved 50 leading research groups analyzing the DNA from 2,000 patients for each of the seven conditions and 3,000 healthy volunteers.
Genome-Wide
They used "gene chips" to scan hundreds of thousands of DNA markers to identify common genetic differences across the whole genome. Many of the genes identified by the team of 200 scientists were in parts of the genome not previously thought to be associated with disease.
In the future it may be possible to test people for combinations of genes to find out their lifetime risk of a disease, which would enable them to modify their lifestyle or undergo screening.
One of the most exciting finds was a previously unknown gene common to type 1 diabetes and Crohn's Disease, a type of inflammatory bowel disorder, suggesting that they share similar biological pathways. The team also unexpectedly found a process known as autophagy -- a process of clearing bacteria from within cells -- is important in the development of Crohn's Disease.
We have taken a big step forward in understanding the genetics underlying some of the most prevalent diseases affecting human health." -- Dr. Mark Walport
And in Type-1 Diabetes, they identified several genetic regions that increase the risk of developing the condition. The WTCCC project has already played a part in recent reports of an obesity gene, three new genes linked to Type-2 Diabetes, and a genetic region on Chromosome 9 associated with coronary heart disease.
Major Advances
Prof. Peter Donnelly, Chairman of the WTCCC and Professor of Statistical Science at Oxford University, said the research was a "new dawn," and they had learned more in the past 12 months than they had in the previous 15 years. "If you think of the genome as very long road that you are trying to find your way along in the dark, previously we have only been able to turn lights on in a small number of places, but now we can turn on lights in a large number of places -- in this case half a million lights." "Many of the most common diseases are very complex, involving both 'nature' and 'nurture' genes interacting with our environment and lifestyles." "By identifying the genes underlying these conditions, our study should enable scientists to understand better how disease occurs, which people are most at risk and, in time, to produce more effective, more personalized treatments."
Dr. Mark Walport, Director of the Wellcome Trust, added that all the data are being made publicly available to ensure that scientists across the globe have immediate access to the results. "We have taken a big step forward in understanding the genetics underlying some of the most prevalent diseases affecting human health."
Matt Hunt, Science Information Manager at Diabetes UK said, "The WTCCC has been an excellent example of collaborative working and has produced some exciting results about the genetics of Type-1 and Type-2 Diabetes."
Ref.:
Nicholas Wade, "Researchers Detect Variations in DNA That Underlie Seven Common
Diseases," The New York Times, p. A30 (June 7, 2007).







May 22, 2007; Dr. Coles presented a talk on "The Medical Consequences of the War in Iraq" to the Optimists International of Pacific Palisades at an 8 o'clock breakfast meeting to an audience of 30 hosted by Mr. Lester Wood and later that day to the USC LA-County Hospital Medical Residents and Students at Noon to an audience of about 20. Dr. Andrew Hendifar, M.D. Chief Resident in Medicine hosted the USC Noon Conference. Click on the PSR website for a close approximation to the PowerPoint slides that were presented.
May 30, 2007; The third photo is for the same talk given to the Hawthorne Rotary Club at the Hacienda Hotel on Sepulveda Blvd. in El Segundo and attended by 17 members.
June 3, 2007; The fourth photo is for the same talk given to the "James Gang" in the Hollywood Hills (actors, writers, directors, producers, journalists in the entertainment industry), hosted by Judy James (bimonthly meetings) on a Sunday afternoon to an audience of 21 members.
June 7, 2007; The fifth photo was taken at the Annual PSR Awards Dinner at the Hilton Hotel in Beverley Hills in which The Hon. Joseph Wilson and his wife Valerie were honored with The Peacemaker Award for services to our country. Recall that Mr. Wilson disabused the American public in a newspaper op-ed piece of the false British intelligence that Saddam Hussein was seeking to purchase "yellow cake" [unenriched Uranium] in the African country of Niger as part of a larger program of WMD (Weapons of Mass Destruction), contrary to what we had been told by the President in his State-of-the-Union Address. This was one of the major justifications for the subsequent war and occupation of Iraq ("regime change"). Also, recall that Joe's wife Valerie was a CIA (covert) Operations Officer whose identity was surreptitiously disclosed to the media by Vice President Dick Cheney's Chief-of-Staff, "Scooter" Libby, who was subsequently charged with a felony and, still expressing no regret, was found guilty and sentenced by the Judge to 30 months in prison and fined $250,000. President Bush will soon be faced with the controversial decision of whether to issue a pardon to Mr. Libby before he leaves office. The Wilson's have filed additional civil lawsuits against the Vice President and against the CIA. Valarie's book discussing this case is planned to be published by Simon and Schuster in October (not yet cited on Amazon.com), while the CIA has thus far blocked publication on the grounds that the book violates national-security laws. Valarie denies that any secret information is revealed, and that all of the delay is politically motivated.
__________________
October 24, 2007;
Refs.:
1. 
Valerie Plame Wilson, Fair Game: My Life as a Spy, My Betrayal by the White House
(320 pages; Simon & Schuster, New York; October 22, 2007; $15.60 on Amazon.com
).
See Ref. 9 below.
2. Tim Rutten, "Now It's Plame's Turn To Dish," The Los Angeles Times, pp. E1-12
(October 23, 2007).
_______________
The Wilsons have recently moved from Washington, D.C. to Santa Fe, NM with their seven-year old twins.
Monday, July 2, 2007; 6:00 PM EDT; Kennebunkport, ME (AP) -- President George W. Bush has commuted Scooter Libby's 30- month prison sentence (without issuing a "full pardon.") Therefore, no matter what the outcome of his appeal, he will never need to serve any jail time following the unanimous three-judge guilty verdict and sentence for the felony conviction for his obstruction of justice in this CIA-leak case. Nevertheless, the President's decision (and subsequent comments by the Vice President) speak volumes about the level of corruption in this Administration, consistent with its obvious disrespect for the law when it applies to itself. Scooter's conviction, monetary fines, and his obligation to be on probation for two years will remain in force. However, there is considerable pent-up anger about this blatantly political decision amongst Democratic presidential candidates, Congressmen, and Senators in the US Congress.
Editorial Comment: This commutation amply demonstrates how Messrs. Bush and Cheney reward blind loyalty among their minions. Furthermore, we remain stunned by this hideous miscarriage of justice. All this, while the President orchestrates well-timed media photo-ops of himself fishing with Russian President Putin in Maine in advance of the 4th-of-July holiday. Because these lawsuits are still pending, we can't predict how Valerie Plame's civil suits against Scooter Libby, the Vice President, the CIA, etc. will move through their legal processes [8]. We're not sure what the capacity of a sitting President might be to interfere in a judicial proceeding that operates in a civil rather than a criminal case, but we suspect that the final chapter of Valerie's book on her life at the CIA is still to be written. The Wilsons and the Bush Administration will probably remain bitter adversaries to the end, unless Joe and Valerie first obtain satisfaction if Mr. Bush were to be impeached for lying to the American people about the reasons for our going to war in Iraq in his State-of-the-Union Address prior to his term running out. If his term were to run out, however, the issue would then become moot. A national office for impeaching both President Bush and Vice President Cheney has been established here in Los Angeles [10].
Refs.:
1. John D. McKinnon, "Sparing Libby Cuts Two Ways," The Wall Street Journal, pp.
A1, 3 (July 3, 2007).
2. Scott Shane and Neil A. Lewis, "Bush Commutes Libby Sentence Saying 30 Months Is
Excessive': Short of Pardon: Reaction Is Polarized, Reflecting Passions of CIA Case," The
New York Times, pp. A1,15 (July 3, 2007).
3. Editorial, "Soft on Crime," The New York Times p. A20 (July 3, 2007).
4. David G. Savage and Richard B. Schmitt, "Bush Spares Libby from Prison: Cheney's Former
Aide Won't Have To Serve His 30-Month Sentence, But He's Not Pardoned in the CIA Leak
Case," The Los Angeles Times, pp. A1,16 (July 3, 2007).
5. Richard B. Schmitt and David G. Savage, "Libby's Sentence Was Not Unusual," The Los
Angeles Times, p. A1,16 (July 4, 2007).
6. Editorial, "Lucky Libby: Martha Stewart, Even Paris Hilton, Served Time. But There's the
Rule of Law -- and the Rules of Bush," The Los Angeles Times, p. A20 (July 4, 2007).
7. Ronald Brownstein, "The Libby Lesson for Iraq," The Los Angeles Times, p. A21
(July 4, 2007).
8. Richard B. Schmitt, "Plame's Suite Against Cheney, Others Dismissed: The ex-CIA Operative
Wanted Them To Pay for Blowing Her Cover," The Los Angeles Times, p. A16 (July
20, 2007).
9. Josh Getlin, "Court Won't Let Plame Reveal Her Years at CIA," The Los Angeles
Times, p. E6 (August 3, 2007).
The memoir entitled Fair Game: My Life As a Spy, My Betrayal by the White House,
will be published in the Fall by Simon and Schuster. The dates of Plame's employment at CIA
before 2002 have already been made public on a Federal website and should no longer be
considered classified information; however, US District Judge, Barbara S. Jones ruled otherwise.
We suspect that when the book is reviewed, this data point will become instantly available
making the adverse ruling moot.
10. Howard Blume, "A Place to Push Impeachment: An Office Opens Near Beverly Center To
Organize Activists Seeking the Ouster of President Bush and Vice President Dick Cheney,"
The Los Angeles Times, p. B3 (July 5, 2007).
US Representative Maxine Waters (D., CA) has lent her support. She is a member of
the House Judiciary Committee that would be needed to bring the charge of impeachment for
"High Crimes and Misdemeanors" against the current Administration. Click for
details if you wish to learn more about the
process of impeachment, circulate petitions, or volunteer your time for this effort.
August 1, 2007; The sixth photo is at the Westchester Rotary International Club Meeting at Noon on August 1st at the LAX Crowne Plaza Hotel to 53 persons.
August 5, 2007; The seventh photo is a talk on "Nuclear Challenges" to ~40 members of the Sepulveda Unitarian Universalist Society of North Hills, CA.
May 16, 2007; Sacramento, CA; Late this afternoon, The California Supreme Court respectfully declined without further explanation to hear the third and final appeal to overturn a lower-court decision brought against The California Institute for Regenerative Medicine (CIRM) by two institutional plaintiffs [ The California Family Bioethics Council, and The Life Legal Defense Foundation] in a lawsuit arguing that Proposition 71 was unconstitutional. Justice Joyce Kennard declined to review the case with no other explanation. Recall that Proposition 71 was passed by California voters in November 2004 by a significant margin. This lawsuit has encumbered the sale of the bonds needed to fund the research for several years now. At last, hopefully, we can get back to doing the work that needs to be done without having to worry about adequate funding. The legal case was known as California Family Bioethics Council vs. California Institute for Regenerative Medicine, S151574.
Mr. Robert Klein, who Co-authored Proposition-71 and serves as Chairman of the Agency's Governing Board, said "The Court's decision frees up the entire $3 billion, providing California with more resources for stem-cell research than any nation in the world [including the United States]." "Still," Klein said, "the Bush Administration's policy of barring any Federal funding for embryonic stem-cell research is blocking facilities in other states from moving forward." "The key message is that California is free from these limitations imposed by the extreme right for ideological reasons, and this nation needs to be free from these unreasonable restraints," he said. "Everyone with a family member suffering from a chronic disease needs the entire nation working on therapies, not just one state."
From The Associated Press [3], California's $3 billion stem-cell agency withstood a last ditch legal challenge by abortion opponents and other critics when the State Supreme Court declined to review a lower-court's ruling that upheld the agency's constitutionality. The decision clears the way for the agency, The California Institute for Regenerative Medicine (CIRM), to dole out billions in research grants that had been held up by litigation.
Ms. Dana Cody, an attorney for the anti-abortion group, The Life Legal Defense Foundation, said, "I don't think that people realize that basically what's happening is fetal farming [catch the fricative alliteration]. You're creating life to experiment with it and ultimately destroy it." [2]
Editorial Remark: I cannot understand how someone like Ms. Cody, who presumably speaks the English language as a native speaker and is sufficiently educated to have passed the California Bar Exam, can get her conceptual semantics so wrong in just two sentences! She doesn't appear to understand the most elementary definitions of the words she uses so casually, but maybe she does. In the case she does, she is intentionally exploiting the ambiguity of the English language for the sake of her own religious agenda. Let me explain: First, we are not proposing to 'farm' [cultivate] anything [ostensibly this was a poetic metaphor]. But more importantly, secondly, we are not dealing with fetuses, but rather with pre-implantation embryos. This is a gross error of argument. Thirdly, we are not creating 'life.' Rather we intend to create human tissues with human DNA, but which are not 'human life.' It is possible for human tissues to be alive but not to be "life." We do not believe that there is anything even remotely like human life at the blastocyst stage of embryonic development, even though the blastocyst can be thought of as alive. The reason for this requires a lengthy explanation of the definition of 'a human person,' with all the legal rights of a living person in the outside world. Fourthly, the alleged human life that we are supposed to be creating is not being created for random 'experimentation' but for experimentation leading to therapy (thus having a legitimate purpose). But more importantly, fifthly, we would not be 'destroying' a human life that anyone has created. We are proposing to harvest the microscopic cells of a blastocyst for therapeutic purposes. Accepting Ms. Cody's premise leads to the seductive anomaly of 'murdering' a blastocyst as a person, something that is completely impossible in principle, unless one accepts the false and ludicrous premise that human personhood begins at conception. But this is the fallacy of confusing an acorn with an oak tree. One is a potential tree. The other is a real tree. By crushing an acorn, one is not, I repeat not, chopping down a large oak tree. That doesn't even make sense, does it? A potential person is not a person. A pre-implantation embryo is not a person. Let's get our facts straight and our words defined properly before we get into any further debate, now that the legal constitutionality arguments have been exhausted.
Refs.:
1.Jason Dearen, Associated Press "California High Court Clears the Way for Stem-Cell
Grants" The San Francisco Chronicle (May 17, 2007).
2. Mary Engel, "Hurdle to Stem Cell Funds Cleared: The State High Court Declines To Hear
Appeals, Opening the Way for the First of $3 Billion in Voter-Approved Bonds To Be Issued by
July," The Los Angeles Times, pp. B1, 12 (May 17, 2007).

May 15, 2007; Mrs. Yvonne Wilson who will be 102 on July 10th, introduced Dr. Stephen Coles to about 50 residents and staff in the Chapel of the Kingsley Manor for a one-hour PowerPoint presentation on Supercentenarians. About six of their 250 residents are about to become centenarians in the coming year.
May 14, 2007; This form of reasoning by analogy, extending Moore's Law to Biology is fraught with peril. For example, who knows how "smart" insects (or mice for that matter) really are. And, how many humans (angels) can you fit on the surface (head) of a compute chip (pin)? Click on the graphic for the complete story from Fortune Magazine and learn such gratuitous facts as "Ray consumes 200 vitamin pills, antioxidants, and other supplements a day!"
Humorous Editorial: The data point at 2050 sure looks like a Singularity, doesn't it? I hope to find you well on that date while we drink champagne together (providing you and I are still here and providing the prophesy is fulfilled), so we can pray collectively to the omniscient computer oracle on Mt. Olympus about how to extend our longevity without biological limit. -- Steve Coles

May 1, 2007; Mandeville, Manchester; JAMAICA; Mary Ewen ("Granny Mary") died today at the alleged age of 128 years old, an age which is virtually certain to be false. Not only that -- her Father was reported to have died at age 130! Click for more details.

April 22, 2007; Anthony Gentry discovered the Chimpanzee named Cheeta (a.k.a. Jiggs) in a Liberian jungle at the age of six months in 1932 [sic]. This month, Cheeta's celebrated his 75th birthday! [sic] Over his career, he appeared in over 50 Hollywood movies. His first screen role, in 1934, was opposite Tarzan, played by Johnny Weissmuller. His last film role was in Dr. Dolittle (1967), after which he retired to a sanctuary in Palm Springs, California. His current trainer, Mr. Daniel Westfall, gives him an insulin shot every day for his diabetes. Chimps in the wild rarely live past 40. In captivity, 50 is considered extreme. Therefore, Cheeta is really a Methuselah among chimps! [sic]
Having recently completed the DNA sequences for the human, the chimp, and the monkey genomes, we now know that humans share 98.77 percent of our genes in common with chimps [3], while only 93 percent of our genes are in common with monkeys [4]. It is still an open question as to how 75 chimp years (Cheeta) compares with 122 human years ( Jeanne Calment), since there is not a simple correspondence between genotypes and a polygenic phenotypes, like longevity, across species boundaries.
Refs.:
1. J. R. Moehringer, "Out of the Hollywood Jungle: Tarzan's Cheeta Turns 75. He Shares His
Secrets on Stardom, Love, and Longevity: Cheeta Speaks: He's Outlived Them All, Tarzan,
Trigger, Ronald Reagan, Now Retired in Palm Springs, the Worlds's Oldest Chimp Gets Some
Stuff Off His Chest," West Magazine, The Los Angeles Times, pp. 12-15, 36
(April 22, 2007).
2. For nine more photos of Cheeta and his artwork click on
latimes.com/cheeta.
3. John Noble Wilford, "Almost Human, and Sometimes Smarter," The New York Times
, p. D1, 4 (April 17, 2007).
4. Rhesus Macaque Genome Sequencing and Analysis Consortium, "Evolutionary and
Biomedical Insights from the Rhesus Macaque Genome," Science, Vol. 316, No.5822,
pp. 222 - 34 (April 13, 2007).
5. Scott Gold, "Whose Life Is It, Anyway?" The Los Angeles Times, pp. B1,5 (February
13, 2009).
Investigative reporting has identified a fraud. It appears that somewhere along the way there was
a "chimp swap," and the Cheeta now in Palm Springs is not the original Cheeta from the early
movies. Therefore, he is an imposter, and no longer the oldest chimp in the world. (Sigh!). It
seems that he was born in ~1960, not 1932!
6. Richard Schickel, "Book Review: Barking Up a Celebrity Chimp's Tree," The Los
Angeles Times, pp. D1,8 (March 19, 2009).
This article doesn't mention the controversy over Cheeta's actual date of birth.

7. Cheeta, the Chimp Me Cheeta: My Life in Hollywood (Ecco: 336 pages; $16.49 on
Amazon.com; 2009).

In this photo provided by Guinness World Records, 114-year-old Edna Parker,
seated Right, of Shelbyville, IN, and Bertha Fry, 113, of Muncie, IN, seated Left, are
honored by Guinness World Records for being the "highest aggregate age of two people
meeting" in a ceremony in Shelbyville on Saturday, April 21, 2007. Indiana Gov. Mitch Daniels,
Left, applauds the pair while Guinness Representative Derek Musso, Right, holds a Certificate
noting the occasion ( AP Photo/Guinness World Records, Tom Strickland).
Saturday, April 21, 2007; Shelbyville, IN (AP and News Sentinel) -- Two of the oldest people in the United States met Saturday. A relative drove Mrs. Bertha Fry, 113, of Muncie, IN to a Shelbyville nursing home to visit Mrs. Edna Parker, who celebrated her 114th birthday on Friday. Members of both families attended the meeting along with Gov. Mitch Daniels, who presented Parker with a Sagamore of the Wabash. The award recognizes Hoosiers who have endeared themselves to the people of Indiana.
A representative of The Guinness Book of World Records also was on hand to confirm the event. The women's combined age is more than that in any other meeting of two humans on record. Together, they accounted for 227 years and 142 days as of Saturday, breaking the previous record of 219 years. "I'm glad you've come," Parker told Fry. "We have a lot to be thankful for."
Parker, born April 20, 1893, became the second-oldest person in the world in January, following the death of Emma Faust Tillman of Hartford, CT, who was 114 and 67 days old when she died, according to the California-based Gerontology Research Group. The only person older than Parker is Yone Minagawa of Japan, who turned 114 on Jan. 4th.
The group lists Fry, born December 1, 1893 as the third-oldest person in the United States and the fifth-oldest in the world. Both Parker and Fry are former school teachers.

April 13, 2007; Scientists from Baylor University in Houston, TX have completed the DNA sequence for a common monkey [1]. For mammals in general, mice, rats, dogs, cows, chimps, and humans have also been sequenced. For other primates, researchers are now beginning to work on decoding the marmoset, the gorilla, the orangutan, and the gibbon. This will surely help us with the search for homologous genes through comparative genomics searching for the phenotype of longevity. The human/chimp lineage diverged from the monkey lineage 25 million years ago, while humans and chimps diverged about 6 million years ago. Humans and the Rhesus Macaque have 93 percent of their genomes in common [2], while humans and chimps have 99 percent of their genomes in common. [3].
Refs.:
1. Rhesus Macaque Genome Sequencing and Analysis Consortium, "Evolutionary and
Biomedical Insights from the Rhesus Macaque Genome," Science, Vol. 316, No.
5822, pp. 222 - 34 (April 13, 2007).
2. Thomas H. Maugh, II, "Macaque Genome Is Decoded," The Los Angeles Times, p.
A17 (April 13, 2007).
3. Lauran Neergaard, "Scientists Decode Rhesus Macaque DNA," Press Telegram, p.
A10 (April 13, 2007).
4. Erika Check, "Make Way for Monkeys: Macaque Genome Sequence Provides Sideways Look
at Human Disease," Nature, Vol. 446, No.7138, p. 840 (April 19, 2007).
5. Michael Hopkin,"Chimps Lead Evolutionary Race: More Chimpanzee Genes Have Been
Positively Selected for Than Human Ones," Nature, Vol. 446, No.7138, p. 841 (April
19, 2007).
6. Susan Milius, "Primate's Progress: Macaque Genome Is Usefully Different," Science
News, Vol. 171, No. 15, p. 227 (April 14, 2007).

After ~20 hours of debate, on Wednesday, April 11, 2007; 6:41 PM EDT;
S.5 (passed)[*] Yea: 63/Nay: 34
S.30 (passed) Yea: 70/Nay: 28
* Three Senators were not present for the vote on S.5; but according to our sources, they would have voted affirmatively, making it one vote shy of a veto override. (If all senators were to vote, it would take a 2/3 majority [>= 67 Yea votes] to override a presidential veto.) Thus, the issue of sustaining the veto is not yet moot, providing only one Senator were to change his or her mind in the right direction. In any event, after a couple more years, when a new President will arrive on the scene, that Congress will presumably revisit this topic once again. Of course, if he or she wished to do so, a new president could change policy by Executive Order.
Refs:
1. "Senate to Begin Debate on Stem-Cell Research," The Wall Street Journal, p. A2
(April 10, 2007).
2. "The Senate Took Up a Bill on Lifting Bush Stem-Cell Funding Curbs..." The Wall Street
Journal, p. A1 (April 11, 2007).
3. "The Senate Voted 63-34 to Lift Bush Curbs..." The Wall Street Journal, p. A1 (April
12, 2007).
4. Nicole Gaouette, "Facing Certain Veto by Bush, Senate Renews Debate on Stem-Cell
Research," The Los Angeles Times, p. A20 (April 11, 2007).
5. Nicole Gaouette, "Senate Strongly Rejects Bush's Curbs on Stem-Cell Research," The Los
Angeles Times, p. A2, 11 (April 12, 2007).
6. Editorial, "A Chance to Change: The Senate's Stem-Cell Vote Gives President Bush an
Opportunity To Alter a Misguided Policy," The Los Angeles Times, p. A20 (April 12,
2007).
7. US Catholic Bishops, "CNA: Reject embryonic research bill, Catholic Bioethics Organization
Urges U.S. Senate
www.catholic.org
(April 12, 2007).
8. Meredith Wadman, "US Senate Passes Stem-Cell Bill -- Again: Bush Promises To Veto
Attempt To Expand Federal Funding," Nature On-Line,
www.nature.com (April 12, 2007).
9. Meredith Wadman, "Stem-Cell Issue Moves Up on the US Agenda: Senate Vote Aims To
Promote Debate," Nature, Vol. 446, No.7138, p. 842 (April 19, 2007).
10. AP "Bush Used a Catholic Prayer Breakfast to Repeat A Veto Threat against a Bill
the Senate Passed This Week To Lift a Stem-Cell Research Funds Curb," The Wall Street
Journal, p. A1 (April 14, 2007).




March 30-31, 2007; Photos taken at the 2007 Edmonton Aging Symposium at The
University of Alberta, CANADA
Click on the left-hand photo above to scroll down and playback any of the individual speaker
presentations (either as Video or as PowerPoint). Speakers with photos are listed in
alphabetical order.




March 28, 2007 [6:30 - 9:00 PM PDT]; Over 100 friends and family members attended the funeral service of Morteza Bahavar, family patriarch born in Iran, age 105, at the Nessah Synagogue in Beverly Hills, CA. At the service, Dr. Stephen Coles was invited to speak for about ten minutes regarding the work of the Supercentenarian Research Foundation. Before Morteza passed away on Thursday morning, his son -- Dr. James Baharvar, M.D. an Internist specializing in Geriatrics in private practice in Santa Monica, CA -- was able to obtain a sample of his father's blood for later DNA analysis, which is now in storage at The Kronos Lab in Phoenix, AZ.

March 27, 2007; Seoul, SOUTH KOREA (Reuters) -- South Korean scientists, once led by disgraced stem-cell researcher Hwang Woo Suk, said on Monday they had created the world's first cloned wolves, which were produced to help an endangered species. A team at Seoul National University, which produced the world's first cloned dog in 2005 -- an Afghan hound named Snuppy -- showed off the two Korean wolves named Snuwolf and Snuwolffy that were born a year and a half ago.
It took the team a while to publish its findings likely because of extra scrutiny due to being implicated in fraud, a member said. "Normally, scientific periodicals would not ask for mtDNA verification, but we needed to produce it due to previous problems," said Prof. Lee Byung-chun, who heads the research team. Lee said the quarterly periodical Cloning and Stem Cells will publish the team's findings in its upcoming issue. Lee said cloning the Korean wolf could help the species survive. "Wolves have not been spotted in the wild in South Korea for about 20 years," Lee said, and the only ones that are known to exist in the South are in a small pack of about ten at a nature park in Seoul.
Snuppy was dubbed one of the most amazing inventions of 2005 by Time magazine. Independent testing has concluded the dog was an actual clone. Hwang and other members have since left their posts at the University after his team fabricated data in two papers on human embryonic stem cells that have since been withdrawn. Hwang is facing trial on fraud, embezzlement, and violating the country's bioethics laws. In December, the team said it had cloned three more Afghan hounds and improved the efficiency of its cloning methods.
For the wolf cloning, it transferred 251 reconstructed embryos to 12 surrogate mothers to produce two living clones. Canines are considered among the most difficult mammals to clone because of their peculiar estrus cycle.
________________
April 20, 2007; We have just learned that there are potential problems with the Cloning and Stem Cells paper submitted by Byung-Cheon Lee and Nam-Sik Shin of Seoul National University (SNU). There is a claim that some of the statistics were manipulated to exaggerate the success rate of the cloning and that a table on DNA sequence-analysis contained errors. Lee admitted to unintentional miscalculations at a recent press conference. In the meantime, the journal has removed the paper from its website pending a further investigation. SNU has submitted for external analysis blood and cell samples from all parties, including the two wolves Snuwolf and Snuwolffy, a dog who supplied the enucleated eggs, and the wolf whose DNA was used by SCNT. Results are expected soon, and we will keep you informed.
Ref.: "Crying Wolf?" Science, Vol. 316, No. 5823, p. 347 (April 20, 2007).
May 4, 2007; We are now pleased to report that DNA evidence has been provided by independent testing to exonerate The Seoul National University (SNU) team, and they did indeed successfully clone their two wolves. To be charitable, they were sloppy in editing their publication accepted in the journal Cloning and Stem Cells (March 2007). There was a simple calculation error in rates and at least one table was mislabeled. The journal, which had retracted the paper pending the investigation, will now post a revised version back on-line as soon as they receive an official report from the University.
Ref.: "Wolf Clones in the Clear," Science, Vol. 316, No. 5825, p. 669 (May 4, 2007).
March 20, 2007; NIH Director Dr. Elias A. Zerhouni, testifying before Sen. Tom Harkin (D. IA) Subcommittee Chairman, surprised everyone with his unequivocal endorsement of embryonic stem-cell research. "Studies claiming that adult stem cells have as much potential as embryonic stem cells do not hold water, scientifically. I think that they are overstated, and we do not know at this point where the breakthrough will come from... all angles in stem-cell research should be pursued." Sen. Harkin responded, "My hope is that the White House gets the message and doesn't continue to stand in the way of progress." Prof. Fred H. Gage of the Salk Institute in La Jolla, CA said, "If this statement puts him [Zerhouni] at risk, all the more reason to respect his judgement." However Wendy Wright President of Concerned Women for America, an opponent of embryonic stem-cell research, said, "He's basically showing his true colors as an advocate and, sadly, that shows he's not looking at the issue objectively."
Refs.:
1. Richardo Alonso-Zaldivar and Karen Kaplan, "Loosening of Stem-Cell Limits Backed: The
NIH Director Calls for Lifting Bush's Federal Funding Restrictions," The Los Angeles
Times, pp. A1,12 (March 20, 2007).
2. "Bush Limits Should be Lifted on Federal Funding of Embryonic Stem-Cell Research,"
The Wall Street Journal, p. A1 (March 20, 2007).
February 27, 2007; San Francisco, CA -- A state appeals court upheld the constitutionality of California's voter-created Stem-Cell Institute Monday, decisively affirming a lower court decision less than two weeks after hearing oral arguments in the case. [1] Robert Klein, Chairman of the California Institute for Regenerative Medicine's oversight committee and the author of the original initiative, said he believed the court's decision would probably clear the way for the sale of bonds and full funding of the $3 billion earmarked for stem-cell research by voters in 2004 [Prop. 71].
"Let me tell you it doesn't get much better than that when the court is making certain that it's 'jealously guarding' the initiative's power," Klein said of the 58-page opinion. "This decision will help us a great deal in trying to achieve our objective of having the Supreme Court refuse to hear an appeal."
The Institute approved 72 research grants totaling $45 million earlier this month, using a state loan arranged by Gov. Arnold Schwarzenegger. Plaintiffs in the consolidated lawsuits People's Advocate, National Tax Limitation Foundation and California Family Bioethics Council may appeal to the state Supreme Court.
"We're going to read the decision carefully, and then we'll consult with our clients," said [Ms.] Dana Cody, Executive Director of Life Legal Defense Foundation, which represents two of the groups that sued. "Chances are we probably will appeal."
Refs.:
1. "Court Upholds Stem-Cell Institute," The Los Angeles Times, p. B3 (February 27,
2007).
2. Andrew Pollack, "California Stem-Cell Research Is Upheld by Appeals Court," The New
York Times, p. A16 (February 27, 2007).
The sale of $3 billion in bonds for CIRM could begin in another four months, if the California
Supreme Court in Sacramento were to "decline to hear the case."
"Ms. Cody is Executive Director of the Life Legal Defense Foundation, an anti-
abortion group, but in this lawsuit is representing two anti-tax groups: The People's
Advocate and The National Tax Limitation Foundation. The third plaintiff is
The California Family Bioethics Council, which is affiliated with Focus on the
Family, a ministry founded by [Dr.] James C. Dobson, [Ph.D.] [located in Colorado
Springs]."
February 27, 2004;
Goran Bjelakovic, MD, DrMedSci; Dimitrinka Nikolova, MA; Lise Lotte Gluud, MD,
DrMedSci; Rosa G. Simonetti, MD; Christian Gluud, MD, DrMedSci
"Mortality in Randomized Trials of Antioxidant Supplements for Primary and Secondary
Prevention: Systematic Review and Meta-analysis"
JAMA, Vol. 297, No. 8, pp. 842-57(February 28, 2007).
Context: Antioxidant supplements are used for prevention of several diseases.
Objective: To assess the effect of antioxidant supplements on mortality in randomized primary and secondary prevention trials.
Data Sources and Trial Selection: We searched electronic databases and bibliographies published by October 2005. All randomized trials involving adults comparing Beta Carotene, Vitamin A, Vitamin C (ascorbic acid), Vitamin E, and selenium either singly or combined vs. placebo or vs. no intervention were included in our analysis. Randomization, blinding, and follow-up were considered markers of bias in the included trials. The effect of antioxidant supplements on all-cause mortality was analyzed with random-effects meta-analyses and reported as relative risk (RR) with 95% confidence intervals (CIs). Meta-regression was used to assess the effect of covariates across the trials.
Data Extraction: We included 68 randomized trials with 232 606 participants (385 publications).
Data Synthesis: When all low- and high-bias risk trials of antioxidant supplements were pooled together there was no significant effect on mortality (RR, 1.02; 95% CI, 0.98-1.06). Multivariate meta-regression analyses showed that low-bias risk trials (RR, 1.16; 95% CI, 1.05-1.29) and Selenium (RR, 0.998; 95% CI, 0.997-0.9995) were significantly associated with mortality. In 47 low-bias trials with 180 938 participants, the antioxidant supplements significantly increased mortality (RR, 1.05; 95% CI, 1.02-1.08). In low-bias risk trials, after exclusion of Selenium trials, Beta Carotene (RR, 1.07; 95% CI, 1.02-1.11), Vitamin A (RR, 1.16; 95% CI, 1.10-1.24), and Vitamin E (RR, 1.04; 95% CI, 1.01-1.07), singly or combined, significantly increased mortality. Vitamin C and Selenium had no significant effect on mortality.
Conclusions: Treatment with Beta Carotene, Vitamin A, and Vitamin E may increase mortality. The potential roles of Vitamin C and Selenium on mortality need further study.
Author Affiliations: The Cochrane Hepato-Biliary Group, Copenhagen Trial Unit, Center for Clinical Intervention Research, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark (Drs. Bjelakovic, L. L. Gluud, Simonetti, and C. Gluud and Ms. Nikolova); Department of Internal Medicine, Gastroenterology and Hepatology, University of Nis, Nis, Serbia (Dr. Bjelakovic); and Divisione di Medicina, Ospedale V. Cervello, Palermo, Italy (Dr. Simonetti).
Refs.:
1. AP Chicago, IL and CNN, "Antioxidant Vitamins A, C, E, Don't
Lead to Longer Life" (February 27, 2007).
2. Tara Parker-Pope, "High Doses of Antioxidants May Hurt More Than Help," pp. A1, D1,8
The Wall Street Journal (February 28, 2007).
"Part of the reason the research is so confusing is that numerous observational studies show that
people who eat diets rich in vegetables, which contain antioxidants, have far better health than
non-vegetable eaters. But in studies of vegetable eaters, the does of antioxidants is typically far
lower than that used in studies of antioxidant supplements. The vast majority of the research
uses high doses of antioxidants to treat unhealthy people, like smokers or persons with existing
heart disease. More study of the health effects of supplements needs to be done on heathy,
younger people." In other words, there's no point in watering the ashes the next day, after a forest
fire has already passed over your home the night before.
3. Thomas H. Maugh, II, "Report Questions Value and Safety of Some Vitamins: Some Dispute
an Analysis of 68 Studies That Finds Antioxidant Supplements Do More Harm Than Good,"
The Los Angeles Times, pp. A2, 10 (February 28, 2007).
4. Tara Parker-Pope, "Health Journal: Vitamin Lowdown: The Good, the Bad, and the
Unknown," The Wall Street Journal, p. D1 (March 6, 2007).
5. "Study Citing Antioxidant Vitamin Risks Based on Flawed Methodology"
(February 27, 2007); Corvallis, OR A study being published today on possible health risks
of antioxidant supplements is based on flawed methodology and ignores the broad totality
of evidence that comes to largely opposite conclusions, experts said today from the Linus
Pauling Institute at Oregon State University.
6. Council for Responsible Nutrition, "Meta-analysis on Antioxidants
Provides Muddled Conclusions: Body of Scientific Research Shows Antioxidants Provide
Benefits for Maintaining Good Health."







February 27, 2007; L. Stephen Coles, M.D. and Doros Platika, M.D., Directors of the Supercentenarian Research Foundation presented to the Jonathan Club founded in 1895 in downtown Los Angeles at 7:30 AM this morning.




The Supercentenarian Research Foundation (SRF) held its first Scientific Advisory Board (SAB) Meeting on the weekend of February 23-25th at the DoubleTree Hotel in Los Angeles near the LAX Airport with the aim of establishing its protocol for sampling the tissues of living Supercentenarians, storing their samples, and analyzing the data. The meeting was attended by 22 scientists, physicians, and researchers with specialties in different aspects of this SRF Central Project.

February 12, 2007; Healthy and viable mice that survive until adulthood have, for the first time, been cloned from adult stem cells. Scientists from Rockefeller University, including Dr. Elaine Fuchs, used cells called keratinocyte stem cells, which represent a new model system for cloning. Keratinocytes come from the skin, making them a particularly attractive stem-cell source because of their ready accessibility. One day, they could be used to tailor therapies, as well as to better understand and treat diseases. Fuchs and her colleague Peter Mombaerts published their laboratories' findings on-line February 12, 2007, in the Proceedings of the National Academy of Sciences.

January 23, 2007 (AP and CNN) -- Say you're a woman who wants to have fertility treatment but can't afford the $[5 - 6],000 cost. What if you could get it for half-price, by agreeing to donate half the eggs you produce for stem-cell research? Interested? British women may get a crack at that deal in a few months, under a plan pursued by Dr. Alison Murdoch of Newcastle University.
This concept, which resembles a strategy sometimes used to get eggs for fertility treatment, is just one of several new efforts to boost the supply of human eggs needed for research. The shortage has triggered an ethical debate on both sides of the Atlantic: Should women be paid for supplying eggs?
Scientists need eggs for a process called therapeutic cloning, which creates stem cells genetically matched to an individual. It may be used someday to create tissue to treat illnesses such as Diabetes and Parkinson's Disease, providing transplant material that's genetically matched to the patient so that it won't be rejected. Therapeutic cloning may also help scientists develop better drug treatments.
The process involves transferring DNA into human eggs and growing them into five-day-old embryos, from which stem cells are harvested. It's not clear just how many eggs scientists need for this research. But it is clear that for a woman, donating eggs is a significant undertaking. By various estimates, a woman can spend [40 - 56] hours in medical offices, being interviewed, counseled, and subjected to a surgical procedure, under sedation, that retrieves eggs from her body. Before that procedure, she takes hormone injections daily for more than a week to stimulate egg development.
Women donate thousands of eggs in the United States every year to help other women have babies. But, they are paid. The American Society of Reproductive Medicine doesn't recommend a figure but says $5,000 or more requires some justification and that $10,000 is too much. (In fact, some ads for eggs offer far more). The medical group also says it's fine to pay women for producing eggs for stem-cell research. But other guidelines and laws on that topic favor just reimbursing women for expenses.
In fact, the compensation question has split American feminists and advocates for reproductive health and rights, said Marcy Darnovsky, Associate Executive Director of the Center for Genetics and Society. One side says offering money beyond reimbursement risks exploiting disadvantaged women by offering undue inducement to participate, while the other side calls that stance "paternalistic," she said. Ethicist Laurie Zoloth of Northwestern University believes that paying compensation could exploit some women. "Women who give eggs to fertility clinics are doing it for the money," she said, and as a society, "we don't ... want the bodies of the poor used for the needs of the wealthy." "You do not see many Full Professors or CEO's selling eggs to secretaries or house-cleaners," she said in an E-mail.
There are ways to guard against exploitation of vulnerable women, said Kathy Hudson, Director of the Genetics and Public Policy Center at Johns Hopkins University. One would be for local boards that oversee research "to make sure that donors are recruited from a wide variety of groups rather than just the economically disadvantaged," she said. And "limits can be set on the number of times any one woman can participate," she said.
Murdoch, who also directs a fertility treatment center in Newcastle upon Tyne, England, said that when her lab asked fertility-clinic patients to donate eggs, it received only 66 over seven months. "That's just not enough," she said. In contrast, if her new plan attracts two women a week -- chosen because they appear likely to produce lots of eggs -- it would provide 20 eggs each week. "That's still not a lot, but the supply should be steady," she said. Her "egg-sharing" plan resembles an arrangement that's used occasionally at fertility clinics. In that plan, a woman shares her eggs and treatment costs with another woman who wants a baby.
Murdoch's group has permission from Britain's Human Fertilization and Embryology Authority to set up the arrangement for stem-cell research. Now it's a question of raising money to finance it. Murdoch said she hopes to start offering the deal to British women in a few months, and that she has already heard from dozens of women eager to participate. Though the HFEA approved Murdoch's plans in July, it has since started gathering public and expert opinions on whether egg sharing should be permitted. "If the consensus is that this is not a good idea, we can change the policy, and rescind the license," said John Paul Maytum, an HFEA spokesman.
Some stem-cell scientists are skirting the debate by finding other sources of eggs. Dr. George Daley of Harvard's Stem-Cell Institute announced in June that he would use eggs originally produced for fertility treatment but which failed to become fertilized. Usually, such eggs are discarded, but women in the fertility program Daley works with must agree to their use in research.
In any case, the need for eggs may only be temporary. They are, in fact, only a tool to reprogram the inserted DNA so that it will drive the development of an early embryo. Scientists hope to learn enough about that reprogramming process to let them take an ordinary cell from a person and use it to produce other kinds of cells, perhaps without going through an embryo stage. "That might happen in ten years," Murdoch estimated. "And then they wouldn't need eggs any more."
Refs.:
1. "Britain Approved Giving Women about $500, or a Discount on Their Fertility-Treatment
Bill, for Donating Eggs for Stem-Cell Research," The Wall Street Journal, p. A1
(February 23, 2007).
2. Britain To Pay Women Who Donate Eggs," The Los Angeles Times, p. A14
(February 24, 2007).
January 16, 2007
Inherited blood disorders could be a thing of the past. Serafini, et al. report on page 129 that Multipotent Adult Progenitor Cells (MAPC's), produced in vitro from bone marrow, have blood-building capacity in immune-deficient mice. The full complement of blood cell types arises from Hematopoietic Stem Cells (HSC's) of the bone marrow. The ability of these cells to both self-renew and to produce daughter cells capable of any hematopoietic fate makes them an attractive resource for cell-replacement therapy for blood and immune disorders. However, as with adult stem cells from other tissues, long-term culturing of these cells has proven difficult.
MAPC's, on the other hand, can divide seemingly endlessly in culture. These cells, which can give rise to multiple cell types, were discovered by chance when Catherine Verfaillie's group was trying to culture another type of adult stem cell. The group now shows that MAPC's can reconstitute hematopoietic compartments in vivo just as well as HSC's. Indeed they can even give rise to HSC's themselves.
The team irradiated mice to knock out their immune cells and then injected traceable MAPC's. MAPC-derived cells were detectable in the bone marrow, spleen, peripheral blood, and lymph nodes of recipient mice and expressed appropriate B, T, and myeloid cell surface markers. Furthermore, MAPC-derived B and T cells were shown to be functional by their production of immunoglobulin and response to T cell receptor stimulus, respectively. The team showed that MAPC's also gave rise to functional long-term HSC's. Transfer of bone marrow cells from the primary recipient mice into new irradiated mice once again led to hematopoietic reconstitution, as did a third round of transfer from these secondary mice to tertiary recipients.
None of the recipient mice in the study developed tumors, even though some of the MAPC's they received were genetically abnormal because of long-term culturing. Genotyping of the recipients' peripheral blood revealed normal karyotypes, leading Verfaillie to suggest that the genetically abnormal MAPC's were somehow cleared by the body.
The risk of tumor development from transplanted Embryonic Stem Cells (ESC's) is an ongoing concern for their use in therapy. The potential of long-term culture followed by tumor-free cell transfer thus gives MAPC's a therapeutic advantage over both ESCs and HSCs.
__________________________________________________________________
MAPC-derived cells (green) restock the lymph nodes of an immunocompromised mouse.
"Hematopoietic Reconstitution by Multipotent Adult
Progenitor Cells: Precursors to Long-Term Hematopoietic Stem Cells"
by
Marta Serafini[1], Scott J. Dylla[3], Masayuki Oki[1], Yves Heremans[1], Jakub Tolar[2],
Yuehua Jiang[1], Shannon M. Buckley[1,4], Beatriz Pelacho[1], Terry C. Burns[1], Sarah
Frommer[1], Derrick J. Rossi[3], David Bryder[3], Angela Panoskaltsis-Mortari[2], Matthew J.
O'Shaughnessy[2], Molly Nelson-Holte[1,4], Gabriel C. Fine[1], Irving L. Weissman[3], Bruce
R. Blazar[2], and Catherine M. Verfaillie[1,4]
The Journal of Experimental Medicine, Vol. 204, No. 1, pp. 129-39 (January 2007).
1. Stem Cell Institute and
2. Cancer Center and
Department of Pediatrics
Division of Hematology, Oncology, Blood, and Marrow Transplant Program
University of Minnesota
Minneapolis, MN 55455; USA
3. Department of Pathology
Institute for Stem Cell Biology and Regenerative Medicine
Stanford University School of Medicine
Stanford, CA 94305; USA
4. Department of Medicine
Stamcel Instituut Leuven
Katholieke Universiteit Leuven
Leuven 3000, BELGIUM
Abstract:
For decades, in vitro expansion of transplantable Hematopoietic Stem Cells (HSC's) has been an elusive goal. Here, we demonstrate that Multipotent Adult Progenitor Cells (MAPC's), isolated from Green Fluorescent Protein (GFP)-transgenic mice and expanded in vitro for >[40 80] population doublings, are capable of multilineage hematopoietic engraftment of immunodeficient mice. Among MAPC-derived GFP+CD45.2+ cells in the bone marrow of engrafted mice, HSC's were present that could radioprotect and reconstitute multilineage hematopoiesis in secondary and tertiary recipients, as well as myeloid and lymphoid hematopoietic progenitor subsets and functional GFP+ MAPC-derived lymphocytes that were functional. Although hematopoietic contribution by MAPCs was comparable to control KTLS HSCs, approximately 103-fold more MAPC's were required for efficient engraftment. Because GFP+ host-derived CD45.1+ cells were not observed, fusion is not likely to account for the generation of HSC's by MAPC's.
Abbreviations used:
eGFP, enhanced GFP;
ESC, Embryonic Stem Cell;
GVHD, Graft- Vs.-Host Disease;
HSC, Hematopoietic Stem Cell;
LT-HSC, long-term repopulating HSC;
MAPC, Multipotent Adult Progenitor Cell;
mESC, mouse ESC;
NOD, nonobese diabetic;
PB, peripheral blood.
M. Serafini and S.J. Dylla contributed equally to this work.
_________________________
1. Reuters, "Study Backs Potential of Adult Stem Cells," The Wall Street Journal, p. D8 (January 16, 2007).
Stem cells taken from bone marrow replenished the radiation-ravaged immune systems and bone marrow of mice and can make brain and liver cells, scientists reported. These so-called adult stem cells can grow almost indefinitely in the lab and have many of the other valued properties of more controversial embryonic stem cells, Catherine Verfaillie of the University of Minnesota and colleagues reported. "The cells not only survived when transplanted but they completely repopulated the blood system of the mice," Dr. Verfaillie said.
Writing in the Journal of Experimental Medicine, the researchers said the findings suggest adult stem cells can be manipulated to regenerate a range of cells and tissues. The cells are called Multipotent Adult Progenitor Cells, or MAPC's, and their discovery in 2001 provoked a furor among opponents of human embryonic stem-cell research, who said the findings proved scientists don't need to experiment on human embryos to regenerate tissue and organs and produce tailored medicine.
Most stem-cell experts, including Dr. Verfaillie, repudiate this argument. "My research has been misused the whole time. There is a huge reason why we have to continue studying embryonic stem cells," she said in a telephone interview. Last week, the House of Representatives voted to expand federal funding of human embryonic-stem-cell research and the Senate is expected to pass the bill. President Bush, who opposes the work, has promised to veto it again, as he did last July.
The White House issued a report saying work such as Dr. Verfaillie's make embryonic-stem-cell research unnecessary.
Dr. Verfaillie and other experts whose work is cited in the report said experiments are needed on all kinds of stem cells to truly understand them, and Federal funding and oversight is the best way to move the promising field forward.
Stem cells are the body's master cells, and depending on how developed they are, they can give rise to all the cells and tissues in an organism, or to a family of cell types.
For instance, neural stem cells can be directed to form various types of nerve cells, while hematopoietic stem cells form blood, immune system and bone marrow cells. Dr. Verfaillie's MAPC's are more powerful than hematopoietic stem cells. "Scientists must now understand that mouse MAPC's can make normal blood, and we need to explore how they do it," said Stanford University's Irving Weissman, who has been skeptical of Dr. Verfaillie's work but who collaborated with her on this study. These cells might help rebuild damaged immune systems, treat blood cancers, and might help transplant patients by "tricking" their immune systems.
2. "Flaws Found in Adult Stem-Cell Study," p. 9D, USA Today (February 27,
2007).
"A groundbreaking adult stem-cell report contains 'significantly flawed' data, and its conclusions
are 'potentially incorrect.' said a panel of scientists. A 2002 Nature mouse study, led by
the University of Minnesota's Catherine Verfaillie, suggest that adult stem cells could be
developed into all sorts of organ tissues for transplants, removing the need for controversial
embryonic stem-cell treatments. However, an expert panel commissioned by the university [of
Minnesota] began investigating the study after New Scientist magazine raised issues
with the data, leading to the withdrawal of some of it earlier this month. Verfaillie told the
Star Tribune of Minneapolis in a story published Friday that the problem was 'an honest
mistake' that did not affect the study's conclusions."
3. Peter Aldhous and Eugene Samuel Reich, "Fresh Questions on Stem-Cell Findings," New
Scientist, Vol. 193, No. 2596, pp. 12-13 (March 24, 2007).
4. Nicholas Wade, "Panel Finds Flawed Data in a Major Stem-Cell Report," The New York
Times, p. A15 (February 28, 2007).
5. Mary Engel, "Stanford Gets $33 Million for Stem-Cell Research," The Los Angeles
Times, p. B8 (February 28, 2007).
6. Constance Holden, "Stem Cells: Data on Adult Stem Cells Questioned" Science, Vol.
315, No. 5816, p. 1207 (March 2, 2007).
7. Constance Holden, "Stem-Cell Results Questioned," Science, Vol. 315, No. 5820,
p. 1779 (March 30, 2007).

"A New Era Begins: Stem Cells Derived from
Amniotic Fluid Show Great Promise in the Lab and May End the Divisive Ethical Debate Once
and for all. Controversy No More? Researchers Are Proving the Efficacy of a New Category of
Stem Cells That May Avoid Ethical Debate"
by
Mary Carmichaelm, Newsweek [1]
January 7-8, 2007 (MS-NBC TV) -- Stem-cell research is divided into two major camps: One focused on cells from adults, the other on the controversial technique that destroys embryos. But important research published Sunday supports the idea of a third way, a new category of stem cells that are readily available, perhaps ethically trouble-free and possibly as powerful and flexible in function as their embryonic counterparts: "amniotic-fluid stem cells," found in both the placenta and the liquid that surrounds growing fetuses.
The cells are "neither embryonic nor adult. They're somewhere in between," says Dr. Anthony Atala, a tissue-engineering specialist at Wake Forest University who led the research team. (The study appears in the journal Nature Biotechnology [2].) The "AFS cells" rival embryonic stem cells in their ability to multiply and transform into many different cell types, and they eventually could be hugely helpful to doctors in treating diseases throughout the body and building new organs in the lab. At the same time, the amniotic cells can be taken easily and harmlessly from the placenta or from pregnant women by amniocentesis -- which gives them the potential to nullify, or at least bridge, the divide in the stem-cell-research debate. One out of every 50 pregnant women undergoes amniocentesis, a procedure that tests the fetus for genetic defects, and about 1 percent of the cells collected by amniocentesis are stem cells. What's more, the stem cells are also found in the placenta, which is thrown away after birth so doctors may obtain them from all infants, not just those subject to amniocentesis.
All of that means the cells come with little "ethical baggage," says David Prentice, a Senior Fellow in life sciences at the Family Research Council, which has a long-standing position against embryonic-stem-cell research. "I'm just pumped up by this," adds Prentice. "It's fantastic."
The AFS cells thrive and divide in the amniotic fluid and placenta throughout the gestation process. Scientists have studied them for several years, but the new research is the first to fully characterize them and demonstrate their potential. "What Dr. Atala has done is to present eloquently, for the first time, the real power that these cells have," says Dr. Roger De Filippo, a Urologist and Tissue Engineer at Childrens Hospital Los Angeles who called the research a "sentinel paper."
Like those from embryos, the AFS cells are pluripotent, or able to transform into fully-grown cells representing each of the three major kinds of tissue found in the body. Using stem cells taken by amniocentesis from 19 pregnant women, Atala and his colleagues were able to create in the lab nerve cells, liver cells, endothelial cells (which line blood vessels) and cells involved in the creation of bone, muscle and fat. (De Filippo's lab has also coaxed amniotic cells into becoming structures found in the kidneys.) Some of the cells in Atala's lab even functioned as they would be expected to in the human body. The liver cells secreted urea, an activity otherwise seen exclusively in their natural counterparts. And, in a development that may hearten patients with Parkinson's Disease and other neurological disorders, the lab's nerve cells secreted Glutamate - a neurotransmitter that is crucial to memory and helps to form Dopamine, which Parkinson's patients lack. The lab also conducted tests on mice with a neurodegenerative disease and showed that the amniotic cells sought out and repopulated damaged areas of the brain.
Amniotic-fluid stem cells share another unique characteristic with embryonic stem cells: They multiply quickly and are remarkably long-lived. The Atala lab's cells divided more than 250 times more than quintuple the life expectancy for stem cells taken from adults. Dr. Dario Fauza, a surgeon at Children's Hospital Boston, says he had achieved "comparable results working with stem cells from amniotic fluid: I practically haven't been able to get them to stop growing." The cells are hardy, a trait that makes them relatively easy to culture. "If you think about where they are in nature, they're floating in the amniotic fluid, in which there is very little oxygen," says Fauza. "So they are very tolerant to low oxygen levels, which makes it easier to manipulate them in the lab."
That resilience may eventually help doctors trying to grow new organs or graft tissue into patients. "When you implant an engineered graft, it's typically vulnerable early on, because it takes a few days for the host to send blood vessels to feed it," says Fauza. "So you need a cell that can take that punishment for a while." You also need, says De Filippo, "a lot of cells to create organs" - a demand that the amniotic cells may meet even more easily than embryonic cells can. In addition, for reasons that are still poorly understood, the amniotic cells do not seem to form the tumors known as teratomas that sometimes arise from embryonic stem cells implanted in animals.
In the short term, Prentice says, the new discovery might not have much legislative impact. "I don't think we're going to see much difference in the rhetoric that both sides will be putting out," he says, particularly in advance of a bill that Congress may vote on next week. But, he adds, "people are becoming more aware that there is another way to get to what we're all after: helping patients, without the ethical concerns and without the bickering." De Filippo also says the new discoveries would be a boon to "the momentum of stem-cell research, especially in California."
Further down the road, the cells could be ideal candidates for "banking," as an increasing number of new parents do today with blood taken from their babies' umbilical cords. Like cord-blood cells, the amniotic cells can be frozen. But once thawed, they live much longer. "The maximum you can do with cord blood cells, which are often used to treat leukemia, is get them to double once," says Atala, compared with the stem cells' lifespan of 250 doublings. A future amniotic stem-cell reserve might be stocked with a variety of genetic types so that cells could be matched to patients with the fewest potential complications.
That era, of course, is well in the future. Many scientists are quick to emphasize that comprehensive human trials are still many years away. It took seven years, Atala notes, just to show the cells' promise, and he declined to estimate how many more it would be before clinical trials could begin, saying, "all those predictions never turn out." There are still many mysteries surrounding amniotic-fluid stem cells -- why they don't cause tumors, why they apparently provoke very little immune response when implanted, and when during embryonic development they first arise -- that might give the FDA pause.
Still, a few experiments on human tissue using cells taken from amniotic fluid are currently in the works. Late last year, a Swiss team reported that it had temporarily been able to grow human heart valves from cells found in amniotic fluid. Dr. Fauza has published a number of large animal studies on tissue engineered from AFS cells over the last several years and is now preparing a clinical trial, this one focusing on children born with a hole in their diaphragms. Babies with the defect today have it patched up with Teflon, "which obviously doesn't grow, so the defect often recurs as the child gets older," says Fauza. Instead, he proposes to construct grafts using amniotic stem cells, and then implant them into newborns. He already has seven years worth of data, all of it encouraging, from performing the same operation on sheep. "The FDA is being helpful, but they are also being very cautious," he says. Still, he hopes the trial will begin in "the not-too-distant future." It's a future that's suddenly looking brighter.
_____________Refs.:
1. Newsweek 2. "Isolation of Amniotic Stem Cell Lines with Potential for Therapy"
Article: Nature Biotechnology, Vol. 25, No. 1, pp. 100-6 (January 2007).
Published on-line: January 7, 2007; | doi:10.1038/nbt1274
Paolo De Coppi[1, 3], Georg Bartsch, Jr.[1, 3], M Minhaj Siddiqui[1], Tao Xu[1],
Cesar C Santos[1], Laura Perin[1], Gustavo Mostoslavsky[2], Ang�line C. Serre[2],
Evan Y. Snyder[2], James J Yoo[1], Mark E Furth[1], Shay Soker[1], and Anthony
Atala[1]
[1]. Wake Forest Institute for Regenerative Medicine
Wake Forest University School of Medicine
Medical Center Blvd.
Winston-Salem, NC 27157-1094; USA
[2]. Children's Hospital and Harvard Medical School
300 Longwood Avenue
Boston, MA 02115; USA
[3]. These authors contributed equally to this work.
Correspondence should be addressed to
Anthony Atala
Abstract:
Stem cells capable of differentiating to multiple lineages may be valuable for therapy. We report the isolation of human and rodent Amniotic Fluid-derived Stem (AFS) cells that express embryonic and adult stem-cell markers. Undifferentiated AFS cells expand extensively without feeders, double in 36 hours and are not tumorigenic. Lines maintained for over 250 population doublings retained long telomeres and a normal karyotype. AFS cells are broadly multipotent. Clonal human lines verified by retroviral marking were induced to differentiate into cell types representing each embryonic germ layer, including cells of adipogenic, osteogenic, myogenic, endothelial, neuronal and hepatic lineages. Examples of differentiated cells derived from human AFS cells and displaying specialized functions include neuronal lineage cells secreting the neurotransmitter L-glutamate or expressing G-protein-gated inwardly rectifying potassium channels, hepatic lineage cells producing urea, and osteogenic lineage cells forming tissue-engineered bone.
3. CNN-TV (Monday, January 8, 2007; 4:45 PM PST; TRT = ~3 min.)
4. Wake Forest Institute for Regenerative
Medicine; Winston-Salem, NC (January 8, 2007).
5. AP, "Stem Cells Discovered in Amniotic Fluid,"
The New York Times (January 8, 2007).
6. "Human Stem Cells Found in Amniotic Fluid"
Reuters Washington, D.C. (January 8, 2007)
7. Karen Kaplan, "Stem Cells in Amniotic Fluid Show Promise: A Study Finds They
Offer Key Therapeutic Benefits But Avoid Controversy," The Los Angeles Times, pp.
A1,14 (January 8, 2006).
8. Carl T. Hall, "Amniotic Fluid a Promising Stem-Cell Source: Political Resistance to
Using Human Embryo Drives Research," San Francisco Chronicle, pp. A1,6 (January 8,
2007).
9. Elizabeth Weise, "Stem Cells Extracted from Amniotic Fluid: Method Could Avoid
Controversy," USA Today, p. 7D (January 8, 2007).
10. AP, "Stem Cells From Amniotic Fluid Could Sidestep Controversy," The
Wall Street Journal (January 8, 2007).
11. Paul Elias, "Stem Cells Discovered in Amniotic Fluid," The Washington Post
(January 8, 2007).
12. Sarah Lueck, "New Science on Stem Cells Draws White House Focus," The Wall
Street Journal, pp. A1, 6 (January 10, 2007).
13. Cristen Brownlee, "Biology: Stem Cells Float in Amniotic Fluid," Science News,
Vol. 171, No. 2, p. 30 (January 13, 2007).
14. Nicholas Confessore, "Spitzer Wants New York To Enter Stem-Cell Race," The New
York Times, p. A27 (January 16, 2007).
New York Governor Elliot Spitzer has called for the passage of a $2 billion 10-year bond
initiative to fund stem-cell research within his state.
15. Andrea Stone, "Each Stored Embryo Is a Stem-Cell Debate," USA Today, pp. 1,2A
(January 30, 2007).
16. Reuters, "Stem Cell Research Guidelines Are Offered," The Los Angeles Times
, p. A14 (February 3, 2007).
The International Society for Stem Cell Research has
prepared a new 15-page set of guidelines for embryonic stem-cell research calling for close
scrutiny of scientists and clear consent from women donating eggs for research. It was prepared
by researchers, ethicists, and legal experts from 14 countries. It prohibits Reproductive
Cloning. It allows for some chimeras (animals seeded with human cells) providing
the research is approved by an Institutional Review Board (IRB). It does not settle the issue
of whether women can be paid for egg donation for research. However, Richard Doerflinger of
the U.S. Conference of Catholic Bishops said metaphorically "It's a recipe guide for how to
prepare the chicken, written by the foxes."


January 8, 2007; The SRF Board of Directors and Steering Committee met in San Francisco over the weekend from Friday afternoon to Sunday evening and elected Dr. Doros Platika, M.D. former CEO of Centigenetics, Inc., as its Chairman and CEO.

Click for News Items from 2006.